Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
Methodology
- Questionnaire Design | Methods, Question Types & Examples

Questionnaire Design | Methods, Question Types & Examples
Published on July 15, 2021 by Pritha Bhandari . Revised on June 22, 2023.
A questionnaire is a list of questions or items used to gather data from respondents about their attitudes, experiences, or opinions. Questionnaires can be used to collect quantitative and/or qualitative information.
Questionnaires are commonly used in market research as well as in the social and health sciences. For example, a company may ask for feedback about a recent customer service experience, or psychology researchers may investigate health risk perceptions using questionnaires.
Table of contents
Questionnaires vs. surveys, questionnaire methods, open-ended vs. closed-ended questions, question wording, question order, step-by-step guide to design, other interesting articles, frequently asked questions about questionnaire design.
A survey is a research method where you collect and analyze data from a group of people. A questionnaire is a specific tool or instrument for collecting the data.
Designing a questionnaire means creating valid and reliable questions that address your research objectives , placing them in a useful order, and selecting an appropriate method for administration.
But designing a questionnaire is only one component of survey research. Survey research also involves defining the population you’re interested in, choosing an appropriate sampling method , administering questionnaires, data cleansing and analysis, and interpretation.
Sampling is important in survey research because you’ll often aim to generalize your results to the population. Gather data from a sample that represents the range of views in the population for externally valid results. There will always be some differences between the population and the sample, but minimizing these will help you avoid several types of research bias , including sampling bias , ascertainment bias , and undercoverage bias .
Here's why students love Scribbr's proofreading services
Discover proofreading & editing
Questionnaires can be self-administered or researcher-administered . Self-administered questionnaires are more common because they are easy to implement and inexpensive, but researcher-administered questionnaires allow deeper insights.
Self-administered questionnaires
Self-administered questionnaires can be delivered online or in paper-and-pen formats, in person or through mail. All questions are standardized so that all respondents receive the same questions with identical wording.
Self-administered questionnaires can be:
- cost-effective
- easy to administer for small and large groups
- anonymous and suitable for sensitive topics
But they may also be:
- unsuitable for people with limited literacy or verbal skills
- susceptible to a nonresponse bias (most people invited may not complete the questionnaire)
- biased towards people who volunteer because impersonal survey requests often go ignored.
Researcher-administered questionnaires
Researcher-administered questionnaires are interviews that take place by phone, in-person, or online between researchers and respondents.
Researcher-administered questionnaires can:
- help you ensure the respondents are representative of your target audience
- allow clarifications of ambiguous or unclear questions and answers
- have high response rates because it’s harder to refuse an interview when personal attention is given to respondents
But researcher-administered questionnaires can be limiting in terms of resources. They are:
- costly and time-consuming to perform
- more difficult to analyze if you have qualitative responses
- likely to contain experimenter bias or demand characteristics
- likely to encourage social desirability bias in responses because of a lack of anonymity
Your questionnaire can include open-ended or closed-ended questions or a combination of both.
Using closed-ended questions limits your responses, while open-ended questions enable a broad range of answers. You’ll need to balance these considerations with your available time and resources.
Closed-ended questions
Closed-ended, or restricted-choice, questions offer respondents a fixed set of choices to select from. Closed-ended questions are best for collecting data on categorical or quantitative variables.
Categorical variables can be nominal or ordinal. Quantitative variables can be interval or ratio. Understanding the type of variable and level of measurement means you can perform appropriate statistical analyses for generalizable results.
Examples of closed-ended questions for different variables
Nominal variables include categories that can’t be ranked, such as race or ethnicity. This includes binary or dichotomous categories.
It’s best to include categories that cover all possible answers and are mutually exclusive. There should be no overlap between response items.
In binary or dichotomous questions, you’ll give respondents only two options to choose from.
White Black or African American American Indian or Alaska Native Asian Native Hawaiian or Other Pacific Islander
Ordinal variables include categories that can be ranked. Consider how wide or narrow a range you’ll include in your response items, and their relevance to your respondents.
Likert scale questions collect ordinal data using rating scales with 5 or 7 points.
When you have four or more Likert-type questions, you can treat the composite data as quantitative data on an interval scale . Intelligence tests, psychological scales, and personality inventories use multiple Likert-type questions to collect interval data.
With interval or ratio scales , you can apply strong statistical hypothesis tests to address your research aims.
Pros and cons of closed-ended questions
Well-designed closed-ended questions are easy to understand and can be answered quickly. However, you might still miss important answers that are relevant to respondents. An incomplete set of response items may force some respondents to pick the closest alternative to their true answer. These types of questions may also miss out on valuable detail.
To solve these problems, you can make questions partially closed-ended, and include an open-ended option where respondents can fill in their own answer.
Open-ended questions
Open-ended, or long-form, questions allow respondents to give answers in their own words. Because there are no restrictions on their choices, respondents can answer in ways that researchers may not have otherwise considered. For example, respondents may want to answer “multiracial” for the question on race rather than selecting from a restricted list.
- How do you feel about open science?
- How would you describe your personality?
- In your opinion, what is the biggest obstacle for productivity in remote work?
Open-ended questions have a few downsides.
They require more time and effort from respondents, which may deter them from completing the questionnaire.
For researchers, understanding and summarizing responses to these questions can take a lot of time and resources. You’ll need to develop a systematic coding scheme to categorize answers, and you may also need to involve other researchers in data analysis for high reliability .
Question wording can influence your respondents’ answers, especially if the language is unclear, ambiguous, or biased. Good questions need to be understood by all respondents in the same way ( reliable ) and measure exactly what you’re interested in ( valid ).
Use clear language
You should design questions with your target audience in mind. Consider their familiarity with your questionnaire topics and language and tailor your questions to them.
For readability and clarity, avoid jargon or overly complex language. Don’t use double negatives because they can be harder to understand.
Use balanced framing
Respondents often answer in different ways depending on the question framing. Positive frames are interpreted as more neutral than negative frames and may encourage more socially desirable answers.
| Positive frame | Negative frame |
|---|---|
| Should protests of pandemic-related restrictions be allowed? | Should protests of pandemic-related restrictions be forbidden? |
Use a mix of both positive and negative frames to avoid research bias , and ensure that your question wording is balanced wherever possible.
Unbalanced questions focus on only one side of an argument. Respondents may be less likely to oppose the question if it is framed in a particular direction. It’s best practice to provide a counter argument within the question as well.
| Unbalanced | Balanced |
|---|---|
| Do you favor…? | Do you favor or oppose…? |
| Do you agree that…? | Do you agree or disagree that…? |
Avoid leading questions
Leading questions guide respondents towards answering in specific ways, even if that’s not how they truly feel, by explicitly or implicitly providing them with extra information.
It’s best to keep your questions short and specific to your topic of interest.
- The average daily work commute in the US takes 54.2 minutes and costs $29 per day. Since 2020, working from home has saved many employees time and money. Do you favor flexible work-from-home policies even after it’s safe to return to offices?
- Experts agree that a well-balanced diet provides sufficient vitamins and minerals, and multivitamins and supplements are not necessary or effective. Do you agree or disagree that multivitamins are helpful for balanced nutrition?
Keep your questions focused
Ask about only one idea at a time and avoid double-barreled questions. Double-barreled questions ask about more than one item at a time, which can confuse respondents.
This question could be difficult to answer for respondents who feel strongly about the right to clean drinking water but not high-speed internet. They might only answer about the topic they feel passionate about or provide a neutral answer instead – but neither of these options capture their true answers.
Instead, you should ask two separate questions to gauge respondents’ opinions.
Strongly Agree Agree Undecided Disagree Strongly Disagree
Do you agree or disagree that the government should be responsible for providing high-speed internet to everyone?
You can organize the questions logically, with a clear progression from simple to complex. Alternatively, you can randomize the question order between respondents.
Logical flow
Using a logical flow to your question order means starting with simple questions, such as behavioral or opinion questions, and ending with more complex, sensitive, or controversial questions.
The question order that you use can significantly affect the responses by priming them in specific directions. Question order effects, or context effects, occur when earlier questions influence the responses to later questions, reducing the validity of your questionnaire.
While demographic questions are usually unaffected by order effects, questions about opinions and attitudes are more susceptible to them.
- How knowledgeable are you about Joe Biden’s executive orders in his first 100 days?
- Are you satisfied or dissatisfied with the way Joe Biden is managing the economy?
- Do you approve or disapprove of the way Joe Biden is handling his job as president?
It’s important to minimize order effects because they can be a source of systematic error or bias in your study.
Randomization
Randomization involves presenting individual respondents with the same questionnaire but with different question orders.
When you use randomization, order effects will be minimized in your dataset. But a randomized order may also make it harder for respondents to process your questionnaire. Some questions may need more cognitive effort, while others are easier to answer, so a random order could require more time or mental capacity for respondents to switch between questions.
Step 1: Define your goals and objectives
The first step of designing a questionnaire is determining your aims.
- What topics or experiences are you studying?
- What specifically do you want to find out?
- Is a self-report questionnaire an appropriate tool for investigating this topic?
Once you’ve specified your research aims, you can operationalize your variables of interest into questionnaire items. Operationalizing concepts means turning them from abstract ideas into concrete measurements. Every question needs to address a defined need and have a clear purpose.
Step 2: Use questions that are suitable for your sample
Create appropriate questions by taking the perspective of your respondents. Consider their language proficiency and available time and energy when designing your questionnaire.
- Are the respondents familiar with the language and terms used in your questions?
- Would any of the questions insult, confuse, or embarrass them?
- Do the response items for any closed-ended questions capture all possible answers?
- Are the response items mutually exclusive?
- Do the respondents have time to respond to open-ended questions?
Consider all possible options for responses to closed-ended questions. From a respondent’s perspective, a lack of response options reflecting their point of view or true answer may make them feel alienated or excluded. In turn, they’ll become disengaged or inattentive to the rest of the questionnaire.
Step 3: Decide on your questionnaire length and question order
Once you have your questions, make sure that the length and order of your questions are appropriate for your sample.
If respondents are not being incentivized or compensated, keep your questionnaire short and easy to answer. Otherwise, your sample may be biased with only highly motivated respondents completing the questionnaire.
Decide on your question order based on your aims and resources. Use a logical flow if your respondents have limited time or if you cannot randomize questions. Randomizing questions helps you avoid bias, but it can take more complex statistical analysis to interpret your data.
Step 4: Pretest your questionnaire
When you have a complete list of questions, you’ll need to pretest it to make sure what you’re asking is always clear and unambiguous. Pretesting helps you catch any errors or points of confusion before performing your study.
Ask friends, classmates, or members of your target audience to complete your questionnaire using the same method you’ll use for your research. Find out if any questions were particularly difficult to answer or if the directions were unclear or inconsistent, and make changes as necessary.
If you have the resources, running a pilot study will help you test the validity and reliability of your questionnaire. A pilot study is a practice run of the full study, and it includes sampling, data collection , and analysis. You can find out whether your procedures are unfeasible or susceptible to bias and make changes in time, but you can’t test a hypothesis with this type of study because it’s usually statistically underpowered .
If you want to know more about statistics , methodology , or research bias , make sure to check out some of our other articles with explanations and examples.
- Student’s t -distribution
- Normal distribution
- Null and Alternative Hypotheses
- Chi square tests
- Confidence interval
- Quartiles & Quantiles
- Cluster sampling
- Stratified sampling
- Data cleansing
- Reproducibility vs Replicability
- Peer review
- Prospective cohort study
Research bias
- Implicit bias
- Cognitive bias
- Placebo effect
- Hawthorne effect
- Hindsight bias
- Affect heuristic
- Social desirability bias
A questionnaire is a data collection tool or instrument, while a survey is an overarching research method that involves collecting and analyzing data from people using questionnaires.
Closed-ended, or restricted-choice, questions offer respondents a fixed set of choices to select from. These questions are easier to answer quickly.
Open-ended or long-form questions allow respondents to answer in their own words. Because there are no restrictions on their choices, respondents can answer in ways that researchers may not have otherwise considered.
A Likert scale is a rating scale that quantitatively assesses opinions, attitudes, or behaviors. It is made up of 4 or more questions that measure a single attitude or trait when response scores are combined.
To use a Likert scale in a survey , you present participants with Likert-type questions or statements, and a continuum of items, usually with 5 or 7 possible responses, to capture their degree of agreement.
You can organize the questions logically, with a clear progression from simple to complex, or randomly between respondents. A logical flow helps respondents process the questionnaire easier and quicker, but it may lead to bias. Randomization can minimize the bias from order effects.
Questionnaires can be self-administered or researcher-administered.
Researcher-administered questionnaires are interviews that take place by phone, in-person, or online between researchers and respondents. You can gain deeper insights by clarifying questions for respondents or asking follow-up questions.
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
Bhandari, P. (2023, June 22). Questionnaire Design | Methods, Question Types & Examples. Scribbr. Retrieved September 18, 2024, from https://www.scribbr.com/methodology/questionnaire/
Is this article helpful?

Pritha Bhandari
Other students also liked, survey research | definition, examples & methods, what is a likert scale | guide & examples, reliability vs. validity in research | difference, types and examples, "i thought ai proofreading was useless but..".
I've been using Scribbr for years now and I know it's a service that won't disappoint. It does a good job spotting mistakes”

Research Methodologies: Research Instruments
- Research Methodology Basics
- Research Instruments
- Types of Research Methodologies
Header Image

Types of Research Instruments
A research instrument is a tool you will use to help you collect, measure and analyze the data you use as part of your research. The choice of research instrument will usually be yours to make as the researcher and will be whichever best suits your methodology.
There are many different research instruments you can use in collecting data for your research:
- Interviews (either as a group or one-on-one). You can carry out interviews in many different ways. For example, your interview can be structured, semi-structured, or unstructured. The difference between them is how formal the set of questions is that is asked of the interviewee. In a group interview, you may choose to ask the interviewees to give you their opinions or perceptions on certain topics.
- Surveys (online or in-person). In survey research, you are posing questions in which you ask for a response from the person taking the survey. You may wish to have either free-answer questions such as essay style questions, or you may wish to use closed questions such as multiple choice. You may even wish to make the survey a mixture of both.
- Focus Groups. Similar to the group interview above, you may wish to ask a focus group to discuss a particular topic or opinion while you make a note of the answers given.
- Observations. This is a good research instrument to use if you are looking into human behaviors. Different ways of researching this include studying the spontaneous behavior of participants in their everyday life, or something more structured. A structured observation is research conducted at a set time and place where researchers observe behavior as planned and agreed upon with participants.
These are the most common ways of carrying out research, but it is really dependent on your needs as a researcher and what approach you think is best to take. It is also possible to combine a number of research instruments if this is necessary and appropriate in answering your research problem.
Data Collection
How to Collect Data for Your Research This article covers different ways of collecting data in preparation for writing a thesis.
- << Previous: Research Methodology Basics
- Next: Types of Research Methodologies >>
- Last Updated: Sep 27, 2022 12:28 PM
- URL: https://paperpile.libguides.com/research-methodologies
- Skip to main content
- Skip to primary sidebar
- Skip to footer
- QuestionPro

- Solutions Industries Gaming Automotive Sports and events Education Government Travel & Hospitality Financial Services Healthcare Cannabis Technology Use Case AskWhy Communities Audience Contactless surveys Mobile LivePolls Member Experience GDPR Positive People Science 360 Feedback Surveys
- Resources Blog eBooks Survey Templates Case Studies Training Help center
Home Market Research
Survey Research: Definition, Examples and Methods

Survey Research is a quantitative research method used for collecting data from a set of respondents. It has been perhaps one of the most used methodologies in the industry for several years due to the multiple benefits and advantages that it has when collecting and analyzing data.
LEARN ABOUT: Behavioral Research
In this article, you will learn everything about survey research, such as types, methods, and examples.
Survey Research Definition
Survey Research is defined as the process of conducting research using surveys that researchers send to survey respondents. The data collected from surveys is then statistically analyzed to draw meaningful research conclusions. In the 21st century, every organization’s eager to understand what their customers think about their products or services and make better business decisions. Researchers can conduct research in multiple ways, but surveys are proven to be one of the most effective and trustworthy research methods. An online survey is a method for extracting information about a significant business matter from an individual or a group of individuals. It consists of structured survey questions that motivate the participants to respond. Creditable survey research can give these businesses access to a vast information bank. Organizations in media, other companies, and even governments rely on survey research to obtain accurate data.
The traditional definition of survey research is a quantitative method for collecting information from a pool of respondents by asking multiple survey questions. This research type includes the recruitment of individuals collection, and analysis of data. It’s useful for researchers who aim to communicate new features or trends to their respondents.
LEARN ABOUT: Level of Analysis Generally, it’s the primary step towards obtaining quick information about mainstream topics and conducting more rigorous and detailed quantitative research methods like surveys/polls or qualitative research methods like focus groups/on-call interviews can follow. There are many situations where researchers can conduct research using a blend of both qualitative and quantitative strategies.
LEARN ABOUT: Survey Sampling
Survey Research Methods
Survey research methods can be derived based on two critical factors: Survey research tool and time involved in conducting research. There are three main survey research methods, divided based on the medium of conducting survey research:
- Online/ Email: Online survey research is one of the most popular survey research methods today. The survey cost involved in online survey research is extremely minimal, and the responses gathered are highly accurate.
- Phone: Survey research conducted over the telephone ( CATI survey ) can be useful in collecting data from a more extensive section of the target population. There are chances that the money invested in phone surveys will be higher than other mediums, and the time required will be higher.
- Face-to-face: Researchers conduct face-to-face in-depth interviews in situations where there is a complicated problem to solve. The response rate for this method is the highest, but it can be costly.
Further, based on the time taken, survey research can be classified into two methods:
- Longitudinal survey research: Longitudinal survey research involves conducting survey research over a continuum of time and spread across years and decades. The data collected using this survey research method from one time period to another is qualitative or quantitative. Respondent behavior, preferences, and attitudes are continuously observed over time to analyze reasons for a change in behavior or preferences. For example, suppose a researcher intends to learn about the eating habits of teenagers. In that case, he/she will follow a sample of teenagers over a considerable period to ensure that the collected information is reliable. Often, cross-sectional survey research follows a longitudinal study .
- Cross-sectional survey research: Researchers conduct a cross-sectional survey to collect insights from a target audience at a particular time interval. This survey research method is implemented in various sectors such as retail, education, healthcare, SME businesses, etc. Cross-sectional studies can either be descriptive or analytical. It is quick and helps researchers collect information in a brief period. Researchers rely on the cross-sectional survey research method in situations where descriptive analysis of a subject is required.
Survey research also is bifurcated according to the sampling methods used to form samples for research: Probability and Non-probability sampling. Every individual in a population should be considered equally to be a part of the survey research sample. Probability sampling is a sampling method in which the researcher chooses the elements based on probability theory. The are various probability research methods, such as simple random sampling , systematic sampling, cluster sampling, stratified random sampling, etc. Non-probability sampling is a sampling method where the researcher uses his/her knowledge and experience to form samples.
LEARN ABOUT: Survey Sample Sizes
The various non-probability sampling techniques are :
- Convenience sampling
- Snowball sampling
- Consecutive sampling
- Judgemental sampling
- Quota sampling
Process of implementing survey research methods:
- Decide survey questions: Brainstorm and put together valid survey questions that are grammatically and logically appropriate. Understanding the objective and expected outcomes of the survey helps a lot. There are many surveys where details of responses are not as important as gaining insights about what customers prefer from the provided options. In such situations, a researcher can include multiple-choice questions or closed-ended questions . Whereas, if researchers need to obtain details about specific issues, they can consist of open-ended questions in the questionnaire. Ideally, the surveys should include a smart balance of open-ended and closed-ended questions. Use survey questions like Likert Scale , Semantic Scale, Net Promoter Score question, etc., to avoid fence-sitting.
LEARN ABOUT: System Usability Scale
- Finalize a target audience: Send out relevant surveys as per the target audience and filter out irrelevant questions as per the requirement. The survey research will be instrumental in case the target population decides on a sample. This way, results can be according to the desired market and be generalized to the entire population.
LEARN ABOUT: Testimonial Questions
- Send out surveys via decided mediums: Distribute the surveys to the target audience and patiently wait for the feedback and comments- this is the most crucial step of the survey research. The survey needs to be scheduled, keeping in mind the nature of the target audience and its regions. Surveys can be conducted via email, embedded in a website, shared via social media, etc., to gain maximum responses.
- Analyze survey results: Analyze the feedback in real-time and identify patterns in the responses which might lead to a much-needed breakthrough for your organization. GAP, TURF Analysis , Conjoint analysis, Cross tabulation, and many such survey feedback analysis methods can be used to spot and shed light on respondent behavior. Use a good survey analysis software . Researchers can use the results to implement corrective measures to improve customer/employee satisfaction.
Reasons to conduct survey research
The most crucial and integral reason for conducting market research using surveys is that you can collect answers regarding specific, essential questions. You can ask these questions in multiple survey formats as per the target audience and the intent of the survey. Before designing a study, every organization must figure out the objective of carrying this out so that the study can be structured, planned, and executed to perfection.
LEARN ABOUT: Research Process Steps
Questions that need to be on your mind while designing a survey are:
- What is the primary aim of conducting the survey?
- How do you plan to utilize the collected survey data?
- What type of decisions do you plan to take based on the points mentioned above?
There are three critical reasons why an organization must conduct survey research.
- Understand respondent behavior to get solutions to your queries: If you’ve carefully curated a survey, the respondents will provide insights about what they like about your organization as well as suggestions for improvement. To motivate them to respond, you must be very vocal about how secure their responses will be and how you will utilize the answers. This will push them to be 100% honest about their feedback, opinions, and comments. Online surveys or mobile surveys have proved their privacy, and due to this, more and more respondents feel free to put forth their feedback through these mediums.
- Present a medium for discussion: A survey can be the perfect platform for respondents to provide criticism or applause for an organization. Important topics like product quality or quality of customer service etc., can be put on the table for discussion. A way you can do it is by including open-ended questions where the respondents can write their thoughts. This will make it easy for you to correlate your survey to what you intend to do with your product or service.
- Strategy for never-ending improvements: An organization can establish the target audience’s attributes from the pilot phase of survey research . Researchers can use the criticism and feedback received from this survey to improve the product/services. Once the company successfully makes the improvements, it can send out another survey to measure the change in feedback keeping the pilot phase the benchmark. By doing this activity, the organization can track what was effectively improved and what still needs improvement.
Survey Research Scales
There are four main scales for the measurement of variables:
- Nominal Scale: A nominal scale associates numbers with variables for mere naming or labeling, and the numbers usually have no other relevance. It is the most basic of the four levels of measurement.
- Ordinal Scale: The ordinal scale has an innate order within the variables along with labels. It establishes the rank between the variables of a scale but not the difference value between the variables.
- Interval Scale: The interval scale is a step ahead in comparison to the other two scales. Along with establishing a rank and name of variables, the scale also makes known the difference between the two variables. The only drawback is that there is no fixed start point of the scale, i.e., the actual zero value is absent.
- Ratio Scale: The ratio scale is the most advanced measurement scale, which has variables that are labeled in order and have a calculated difference between variables. In addition to what interval scale orders, this scale has a fixed starting point, i.e., the actual zero value is present.
Benefits of survey research
In case survey research is used for all the right purposes and is implemented properly, marketers can benefit by gaining useful, trustworthy data that they can use to better the ROI of the organization.
Other benefits of survey research are:
- Minimum investment: Mobile surveys and online surveys have minimal finance invested per respondent. Even with the gifts and other incentives provided to the people who participate in the study, online surveys are extremely economical compared to paper-based surveys.
- Versatile sources for response collection: You can conduct surveys via various mediums like online and mobile surveys. You can further classify them into qualitative mediums like focus groups , and interviews and quantitative mediums like customer-centric surveys. Due to the offline survey response collection option, researchers can conduct surveys in remote areas with limited internet connectivity. This can make data collection and analysis more convenient and extensive.
- Reliable for respondents: Surveys are extremely secure as the respondent details and responses are kept safeguarded. This anonymity makes respondents answer the survey questions candidly and with absolute honesty. An organization seeking to receive explicit responses for its survey research must mention that it will be confidential.
Survey research design
Researchers implement a survey research design in cases where there is a limited cost involved and there is a need to access details easily. This method is often used by small and large organizations to understand and analyze new trends, market demands, and opinions. Collecting information through tactfully designed survey research can be much more effective and productive than a casually conducted survey.
There are five stages of survey research design:
- Decide an aim of the research: There can be multiple reasons for a researcher to conduct a survey, but they need to decide a purpose for the research. This is the primary stage of survey research as it can mold the entire path of a survey, impacting its results.
- Filter the sample from target population: Who to target? is an essential question that a researcher should answer and keep in mind while conducting research. The precision of the results is driven by who the members of a sample are and how useful their opinions are. The quality of respondents in a sample is essential for the results received for research and not the quantity. If a researcher seeks to understand whether a product feature will work well with their target market, he/she can conduct survey research with a group of market experts for that product or technology.
- Zero-in on a survey method: Many qualitative and quantitative research methods can be discussed and decided. Focus groups, online interviews, surveys, polls, questionnaires, etc. can be carried out with a pre-decided sample of individuals.
- Design the questionnaire: What will the content of the survey be? A researcher is required to answer this question to be able to design it effectively. What will the content of the cover letter be? Or what are the survey questions of this questionnaire? Understand the target market thoroughly to create a questionnaire that targets a sample to gain insights about a survey research topic.
- Send out surveys and analyze results: Once the researcher decides on which questions to include in a study, they can send it across to the selected sample . Answers obtained from this survey can be analyzed to make product-related or marketing-related decisions.
Survey examples: 10 tips to design the perfect research survey
Picking the right survey design can be the key to gaining the information you need to make crucial decisions for all your research. It is essential to choose the right topic, choose the right question types, and pick a corresponding design. If this is your first time creating a survey, it can seem like an intimidating task. But with QuestionPro, each step of the process is made simple and easy.
Below are 10 Tips To Design The Perfect Research Survey:
- Set your SMART goals: Before conducting any market research or creating a particular plan, set your SMART Goals . What is that you want to achieve with the survey? How will you measure it promptly, and what are the results you are expecting?
- Choose the right questions: Designing a survey can be a tricky task. Asking the right questions may help you get the answers you are looking for and ease the task of analyzing. So, always choose those specific questions – relevant to your research.
- Begin your survey with a generalized question: Preferably, start your survey with a general question to understand whether the respondent uses the product or not. That also provides an excellent base and intro for your survey.
- Enhance your survey: Choose the best, most relevant, 15-20 questions. Frame each question as a different question type based on the kind of answer you would like to gather from each. Create a survey using different types of questions such as multiple-choice, rating scale, open-ended, etc. Look at more survey examples and four measurement scales every researcher should remember.
- Prepare yes/no questions: You may also want to use yes/no questions to separate people or branch them into groups of those who “have purchased” and those who “have not yet purchased” your products or services. Once you separate them, you can ask them different questions.
- Test all electronic devices: It becomes effortless to distribute your surveys if respondents can answer them on different electronic devices like mobiles, tablets, etc. Once you have created your survey, it’s time to TEST. You can also make any corrections if needed at this stage.
- Distribute your survey: Once your survey is ready, it is time to share and distribute it to the right audience. You can share handouts and share them via email, social media, and other industry-related offline/online communities.
- Collect and analyze responses: After distributing your survey, it is time to gather all responses. Make sure you store your results in a particular document or an Excel sheet with all the necessary categories mentioned so that you don’t lose your data. Remember, this is the most crucial stage. Segregate your responses based on demographics, psychographics, and behavior. This is because, as a researcher, you must know where your responses are coming from. It will help you to analyze, predict decisions, and help write the summary report.
- Prepare your summary report: Now is the time to share your analysis. At this stage, you should mention all the responses gathered from a survey in a fixed format. Also, the reader/customer must get clarity about your goal, which you were trying to gain from the study. Questions such as – whether the product or service has been used/preferred or not. Do respondents prefer some other product to another? Any recommendations?
Having a tool that helps you carry out all the necessary steps to carry out this type of study is a vital part of any project. At QuestionPro, we have helped more than 10,000 clients around the world to carry out data collection in a simple and effective way, in addition to offering a wide range of solutions to take advantage of this data in the best possible way.
From dashboards, advanced analysis tools, automation, and dedicated functions, in QuestionPro, you will find everything you need to execute your research projects effectively. Uncover insights that matter the most!
MORE LIKE THIS

QuestionPro: Leading the Charge in Customer Journey Management and Voice of the Customer Platforms
Sep 17, 2024

What is Driver Analysis? Importance and Best Practices

Was The Experience Memorable? (Part II) — Tuesday CX Thoughts

Data Discovery: What it is, Importance, Process + Use Cases
Sep 16, 2024
Other categories
- Academic Research
- Artificial Intelligence
- Assessments
- Brand Awareness
- Case Studies
- Communities
- Consumer Insights
- Customer effort score
- Customer Engagement
- Customer Experience
- Customer Loyalty
- Customer Research
- Customer Satisfaction
- Employee Benefits
- Employee Engagement
- Employee Retention
- Friday Five
- General Data Protection Regulation
- Insights Hub
- Life@QuestionPro
- Market Research
- Mobile diaries
- Mobile Surveys
- New Features
- Online Communities
- Question Types
- Questionnaire
- QuestionPro Products
- Release Notes
- Research Tools and Apps
- Revenue at Risk
- Survey Templates
- Training Tips
- Tuesday CX Thoughts (TCXT)
- Uncategorized
- What’s Coming Up
- Workforce Intelligence
Root out friction in every digital experience, super-charge conversion rates, and optimize digital self-service
Uncover insights from any interaction, deliver AI-powered agent coaching, and reduce cost to serve
Increase revenue and loyalty with real-time insights and recommendations delivered to teams on the ground
Know how your people feel and empower managers to improve employee engagement, productivity, and retention
Take action in the moments that matter most along the employee journey and drive bottom line growth
Whatever they’re saying, wherever they’re saying it, know exactly what’s going on with your people
Get faster, richer insights with qual and quant tools that make powerful market research available to everyone
Run concept tests, pricing studies, prototyping + more with fast, powerful studies designed by UX research experts
Track your brand performance 24/7 and act quickly to respond to opportunities and challenges in your market
Explore the platform powering Experience Management
- Free Account
- Product Demos
- For Digital
- For Customer Care
- For Human Resources
- For Researchers
- Financial Services
- All Industries
Popular Use Cases
- Customer Experience
- Employee Experience
- Net Promoter Score
- Voice of Customer
- Customer Success Hub
- Product Documentation
- Training & Certification
- XM Institute
- Popular Resources
- Customer Stories
- Artificial Intelligence
- Market Research
- Partnerships
- Marketplace
The annual gathering of the experience leaders at the world’s iconic brands building breakthrough business results, live in Salt Lake City.
- English/AU & NZ
- Español/Europa
- Español/América Latina
- Português Brasileiro
- REQUEST DEMO
- Experience Management
- What is a survey?
- Survey Research
Try Qualtrics for free
What is survey research.
15 min read Find out everything you need to know about survey research, from what it is and how it works to the different methods and tools you can use to ensure you’re successful.
Survey research is the process of collecting data from a predefined group (e.g. customers or potential customers) with the ultimate goal of uncovering insights about your products, services, or brand overall .
As a quantitative data collection method, survey research can provide you with a goldmine of information that can inform crucial business and product decisions. But survey research needs careful planning and execution to get the results you want.
So if you’re thinking about using surveys to carry out research, read on.
Get started with our free survey maker tool
Types of survey research
Calling these methods ‘survey research’ slightly underplays the complexity of this type of information gathering. From the expertise required to carry out each activity to the analysis of the data and its eventual application, a considerable amount of effort is required.
As for how you can carry out your research, there are several options to choose from — face-to-face interviews, telephone surveys, focus groups (though more interviews than surveys), online surveys , and panel surveys.
Typically, the survey method you choose will largely be guided by who you want to survey, the size of your sample , your budget, and the type of information you’re hoping to gather.
Here are a few of the most-used survey types:
Face-to-face interviews
Before technology made it possible to conduct research using online surveys, telephone, and mail were the most popular methods for survey research. However face-to-face interviews were considered the gold standard — the only reason they weren’t as popular was due to their highly prohibitive costs.
When it came to face-to-face interviews, organizations would use highly trained researchers who knew when to probe or follow up on vague or problematic answers. They also knew when to offer assistance to respondents when they seemed to be struggling. The result was that these interviewers could get sample members to participate and engage in surveys in the most effective way possible, leading to higher response rates and better quality data.
Telephone surveys
While phone surveys have been popular in the past, particularly for measuring general consumer behavior or beliefs, response rates have been declining since the 1990s .
Phone surveys are usually conducted using a random dialing system and software that a researcher can use to record responses.
This method is beneficial when you want to survey a large population but don’t have the resources to conduct face-to-face research surveys or run focus groups, or want to ask multiple-choice and open-ended questions .
The downsides are they can: take a long time to complete depending on the response rate, and you may have to do a lot of cold-calling to get the information you need.
You also run the risk of respondents not being completely honest . Instead, they’ll answer your survey questions quickly just to get off the phone.
Focus groups (interviews — not surveys)
Focus groups are a separate qualitative methodology rather than surveys — even though they’re often bunched together. They’re normally used for survey pretesting and designing , but they’re also a great way to generate opinions and data from a diverse range of people.
Focus groups involve putting a cohort of demographically or socially diverse people in a room with a moderator and engaging them in a discussion on a particular topic, such as your product, brand, or service.
They remain a highly popular method for market research , but they’re expensive and require a lot of administration to conduct and analyze the data properly.
You also run the risk of more dominant members of the group taking over the discussion and swaying the opinions of other people — potentially providing you with unreliable data.
Online surveys
Online surveys have become one of the most popular survey methods due to being cost-effective, enabling researchers to accurately survey a large population quickly.
Online surveys can essentially be used by anyone for any research purpose – we’ve all seen the increasing popularity of polls on social media (although these are not scientific).
Using an online survey allows you to ask a series of different question types and collect data instantly that’s easy to analyze with the right software.
There are also several methods for running and distributing online surveys that allow you to get your questionnaire in front of a large population at a fraction of the cost of face-to-face interviews or focus groups.
This is particularly true when it comes to mobile surveys as most people with a smartphone can access them online.
However, you have to be aware of the potential dangers of using online surveys, particularly when it comes to the survey respondents. The biggest risk is because online surveys require access to a computer or mobile device to complete, they could exclude elderly members of the population who don’t have access to the technology — or don’t know how to use it.
It could also exclude those from poorer socio-economic backgrounds who can’t afford a computer or consistent internet access. This could mean the data collected is more biased towards a certain group and can lead to less accurate data when you’re looking for a representative population sample.
When it comes to surveys, every voice matters.
Free eBook: Learn how to increase response rates to your survey
Panel surveys
A panel survey involves recruiting respondents who have specifically signed up to answer questionnaires and who are put on a list by a research company. This could be a workforce of a small company or a major subset of a national population. Usually, these groups are carefully selected so that they represent a sample of your target population — giving you balance across criteria such as age, gender, background, and so on.
Panel surveys give you access to the respondents you need and are usually provided by the research company in question. As a result, it’s much easier to get access to the right audiences as you just need to tell the research company your criteria. They’ll then determine the right panels to use to answer your questionnaire.
However, there are downsides. The main one being that if the research company offers its panels incentives, e.g. discounts, coupons, money — respondents may answer a lot of questionnaires just for the benefits.
This might mean they rush through your survey without providing considered and truthful answers. As a consequence, this can damage the credibility of your data and potentially ruin your analyses.
What are the benefits of using survey research?
Depending on the research method you use, there are lots of benefits to conducting survey research for data collection. Here, we cover a few:
1. They’re relatively easy to do
Most research surveys are easy to set up, administer and analyze. As long as the planning and survey design is thorough and you target the right audience , the data collection is usually straightforward regardless of which survey type you use.
2. They can be cost effective
Survey research can be relatively cheap depending on the type of survey you use.
Generally, qualitative research methods that require access to people in person or over the phone are more expensive and require more administration.
Online surveys or mobile surveys are often more cost-effective for market research and can give you access to the global population for a fraction of the cost.
3. You can collect data from a large sample
Again, depending on the type of survey, you can obtain survey results from an entire population at a relatively low price. You can also administer a large variety of survey types to fit the project you’re running.
4. You can use survey software to analyze results immediately
Using survey software, you can use advanced statistical analysis techniques to gain insights into your responses immediately.
Analysis can be conducted using a variety of parameters to determine the validity and reliability of your survey data at scale.
5. Surveys can collect any type of data
While most people view surveys as a quantitative research method, they can just as easily be adapted to gain qualitative information by simply including open-ended questions or conducting interviews face to face.
How to measure concepts with survey questions
While surveys are a great way to obtain data, that data on its own is useless unless it can be analyzed and developed into actionable insights.
The easiest, and most effective way to measure survey results, is to use a dedicated research tool that puts all of your survey results into one place.
When it comes to survey measurement, there are four measurement types to be aware of that will determine how you treat your different survey results:
Nominal scale
With a nominal scale , you can only keep track of how many respondents chose each option from a question, and which response generated the most selections.
An example of this would be simply asking a responder to choose a product or brand from a list.
You could find out which brand was chosen the most but have no insight as to why.
Ordinal scale
Ordinal scales are used to judge an order of preference. They do provide some level of quantitative value because you’re asking responders to choose a preference of one option over another.
Ratio scale
Ratio scales can be used to judge the order and difference between responses. For example, asking respondents how much they spend on their weekly shopping on average.
Interval scale
In an interval scale, values are lined up in order with a meaningful difference between the two values — for example, measuring temperature or measuring a credit score between one value and another.
Step by step: How to conduct surveys and collect data
Conducting a survey and collecting data is relatively straightforward, but it does require some careful planning and design to ensure it results in reliable data.
Step 1 – Define your objectives
What do you want to learn from the survey? How is the data going to help you? Having a hypothesis or series of assumptions about survey responses will allow you to create the right questions to test them.
Step 2 – Create your survey questions
Once you’ve got your hypotheses or assumptions, write out the questions you need answering to test your theories or beliefs. Be wary about framing questions that could lead respondents or inadvertently create biased responses .
Step 3 – Choose your question types
Your survey should include a variety of question types and should aim to obtain quantitative data with some qualitative responses from open-ended questions. Using a mix of questions (simple Yes/ No, multiple-choice, rank in order, etc) not only increases the reliability of your data but also reduces survey fatigue and respondents simply answering questions quickly without thinking.
Step 4 – Test your questions
Before sending your questionnaire out, you should test it (e.g. have a random internal group do the survey) and carry out A/B tests to ensure you’ll gain accurate responses.
Step 5 – Choose your target and send out the survey
Depending on your objectives, you might want to target the general population with your survey or a specific segment of the population. Once you’ve narrowed down who you want to target, it’s time to send out the survey.
After you’ve deployed the survey, keep an eye on the response rate to ensure you’re getting the number you expected. If your response rate is low, you might need to send the survey out to a second group to obtain a large enough sample — or do some troubleshooting to work out why your response rates are so low. This could be down to your questions, delivery method, selected sample, or otherwise.
Step 6 – Analyze results and draw conclusions
Once you’ve got your results back, it’s time for the fun part.
Break down your survey responses using the parameters you’ve set in your objectives and analyze the data to compare to your original assumptions. At this stage, a research tool or software can make the analysis a lot easier — and that’s somewhere Qualtrics can help.
Get reliable insights with survey software from Qualtrics
Gaining feedback from customers and leads is critical for any business, data gathered from surveys can prove invaluable for understanding your products and your market position, and with survey software from Qualtrics, it couldn’t be easier.
Used by more than 13,000 brands and supporting more than 1 billion surveys a year, Qualtrics empowers everyone in your organization to gather insights and take action. No coding required — and your data is housed in one system.
Get feedback from more than 125 sources on a single platform and view and measure your data in one place to create actionable insights and gain a deeper understanding of your target customers .
Automatically run complex text and statistical analysis to uncover exactly what your survey data is telling you, so you can react in real-time and make smarter decisions.
We can help you with survey management, too. From designing your survey and finding your target respondents to getting your survey in the field and reporting back on the results, we can help you every step of the way.
And for expert market researchers and survey designers, Qualtrics features custom programming to give you total flexibility over question types, survey design, embedded data, and other variables.
No matter what type of survey you want to run, what target audience you want to reach, or what assumptions you want to test or answers you want to uncover, we’ll help you design, deploy and analyze your survey with our team of experts.
Ready to find out more about Qualtrics CoreXM?
Get started with our free survey maker tool today
Related resources
Survey bias types 24 min read, post event survey questions 10 min read, best survey software 16 min read, close-ended questions 7 min read, survey vs questionnaire 12 min read, response bias 13 min read, double barreled question 11 min read, request demo.
Ready to learn more about Qualtrics?
Occupational Science & Occupational Therapy
- Home / Help
- OT ejournals
About Research Instruments
Databases for finding research instruments, find research instruments in instrument databases, find research instruments in literature databases.
- Scoping Reviews
- Health Statistics and Data
- Writing Resources
- Transformative Agreements / Read and Publish
- Anatomy Resources
- Step 1: ASK
- Step 2: ACQUIRE/ACCESS
- Step 3: APPRAISE
- Steps 4 & 5: APPLY & ASSESS
- Mobile Apps
- NBCOT Exam Prep
- Library Instruction and Tutorials
- Research instruments are measurement tools, such as questionnaires, scales, and surveys, that researchers use to measure variables in research studies.
- In most cases, it is better to use a previously validated instrument rather than create one from scratch.
- Always evaluate instruments for relevancy, validity, and reliability.
- Many older yet relevant, valid and reliable instruments are still popular today. It is time consuming and costly to validate instruments, so re-using instruments is common and helpful for connecting your study with an existing body of research.
- Although you can conduct an internet search to find research instruments on publisher and organization websites, library databases are usually the best resources for identifying relevant, validated and reliable research instruments.
- Locating instruments takes time and requires you to follow multiple references until you reach the source.
- Databases provide information about instruments, but they do not provide access to the instruments themselves.
- In most cases, to access and use the actual instruments, you must contact the author or purchase the instrument from the publisher.
- In many cases, you will have to pay a fee to use the instrument.
- Even if the full instrument is freely available, you should contact the owner for permission to use and for any instructions and training necessary to use the instrument properly.
- CINAHL Complete This link opens in a new window Most comprehensive database of full-text for nursing & allied health journals from 1937 to present. Includes access to scholarly journal articles, dissertations, magazines, pamphlets, evidence-based care sheets, books, and research instruments.
- Health and Psychosocial Instruments (HAPI) This link opens in a new window Locate measurement instruments such as surveys, questionnaires, tests, surveys, coding schemes, checklists, rating scales, vignettes, etc. Scope includes medicine, nursing, public health , psychology, social work, communication, sociology, etc.
- Mental Measurements Yearbook (MMY) This link opens in a new window Use MMY to find REVIEWS of testing instruments. Actual test instruments are NOT provided. Most reviews discuss validity and reliability of tool. To purchase or obtain the actual test materials, you will need to contact the test publisher(s).
- PsycINFO This link opens in a new window Abstract and citation database of scholarly literature in psychological, social, behavioral, and health sciences. Includes journal articles, books, reports, theses, and dissertations from 1806 to present.
- PsycTESTS This link opens in a new window PsycTESTS is a research database that provides access to psychological tests, measures, scales, surveys, and other assessments as well as descriptive information about the test and its development. Records also discuss reliability and validity of the tool. Some records include full-text of the test.
- Rehabilitation Measures Database RMD is a web-based, searchable database of assessment instruments designed to help clinicians and researchers select appropriate measures for screening, monitoring patient progress, and assessing outcomes in rehabilitation. more... less... This database allows clinicians and researchers to search for instruments using a word search or specific characteristics of an instrument, such as the area of assessment, diagnosis, length of test, and cost. The search function returns relevant results and provides the user with the ability to refine the search. The instruments listed in the database are described with specific details regarding their reliability, validity, mode of administration, cost, and equipment required. Additionally, information to support the user in interpreting the results, such as minimal detectable change scores, cut-offs, and normative values are included. A sample copy of the instrument is also provided when available.
- << Previous: OT ejournals
- Next: Scoping Reviews >>
- Last Updated: Sep 13, 2024 9:49 AM
- URL: https://libguides.usc.edu/healthsciences/ot

- UVM Libraries
- UVM Libraries Research Guides
- Dana Health Sciences Library
Research Instruments: Surveys, Questionnaires, and other Measurement Tools
- Descriptions and Search Tips
| Resources: Descriptions and Search Tips | ||
|---|---|---|
| Sources | Description | Search Tips |
| indexes Nursing and Allied Health literature and provides the detailed descriptions of over 350 popular instruments. | Search by instrument name or acronym and limit to the Publication Type: Questionnaire/Scale [documents with partial or complete examples of questionnaires or scales] or Research Instrument [special CINAHL records that describe research instruments and may have an instrument appended]; | |
| (Education Resources Information Center) | largest database of education literature in the world; includes a variety of document types including journal articles, research reports, curriculum and teaching guides, conference papers, and books
| Search by instrument name or acronym and limit to Publication Type: Tests/Questionnaires [articles where an instrument is included] |
| database of descriptive information for over 20,000 tests from varying fields with an emphasis on education | Search by instrument name Search by keyword in SubjectSearch | |
| comprehensive bibliographic coverage of a wide variety of evaluation and measurement tools for health and psychosocial studies, includes instruments not commercially or widely available | Search by instrument name or acronym in the TI or AC field. Select Primary Source in search options under Source Code. View individual records to see the source reference. Search by keyword in the Measure field Search by instrument name or acronym and select the Source Code: Review Source | |
| describes more than 2,000 standardized English-language tests that assess personality, aptitude, achievement, intelligence, and other neuropsychological behavior. Only the descriptive material and reviews are available full-text. The tests are available for purchase. Coverage is 1989 - present. | Search by instrument name or acronym Search by keyword Click on the Has Review box Limit | |
| public repository of information on evidence-based care quality measures and measure sets, sponsored by the Agency for Healthcare Research and Quality (AHRQ) | Search by keyword or click on Measures to browse by category; use Advanced Search to limit by age, population, etc. | |
| comprehensive index to dissertations and theses from all fields of study, 1861-present. Full text of any dissertations not online is available through interlibrary loan. | Search by instrument name or by variable. Check the appendices of dissertations for the full-text instruments used. | |
| produced by the American Psychological Corporation, index of citations to the international literature of psychology | Search by instrument name or acronym in one search box; in another search box type in the word ‘appended’ and select the Tests and Measures field. OR Search by keyword in the Tests and Measures field Search by instrument name in the Tests & Measures or Keyword or Abstract fields; add additional thesauraus terms such as Test Validity, Test Reliability in the Subjects field | |
| provides access to primarily unpublished tests developed by researchers. All records include a summary of the test as well as the history of its development. What sets PsycTESTS apart is, in many cases, the inclusion of the actual testing instrument. | Search by instrument name or acronym Search by keyword Limit to Record Type Test Review | |
| biomedical database including MEDLINE | Search by instrument name or acronym Search by keyword Use keywords such as validity, reliability or use MeSH terms such as validation studies[pt], validation studies as topic[mh], reproducibility of results[mh] | |
| "All of the surveys and tools from RAND Health are public documents, available without charge (for non-commercial purposes). Some materials listed are not available from RAND Health. Links will take you to other websites, where you will find instructions for use." | Click on Surveys, browse by categories such as quality of life | "Curated instruments have been approved for inclusion by the REDCap Library Oversight Committee (REDLOC) after review for research relevance, accuracy in coding and function, and copyright issues." | Click on the Library tab to see a list of instruments and measures. Full text of instrument is included. | -->
| bibliographic database of over 1,800 journals, conference papers, dissertations, book reviews and selected sociology books on sociology, social welfare, social planning and development. | Search by instrument name and additional terms such as Measures (Instruments) in the Subject Headings field Search by keyword and additional terms such as Measures (Instruments) in the Subject Headings field OR questionnaire*, test*, scale*, measure*, etc. in the Identifier field Search by instrument name or acronym and additional thesaurus terms such as reliability, validity, etc. in the Subject Heading field | |
| database of over 225 questionnaires and interviews intended to assist treatment providers and researchers with screening and assessment of substance use and substance use disorders. Measures that are widely used and have proven reliability and validity are noted with a gold star. | Search or browse by instrument name or acronym; view full records to see the Use & Availability and Copy of Instrument fields Search by variable Search or browse by instrument name or acronym; brief synopsis of reliability and validity included; references provided | |
| multidisciplinary databases covering journals in the sciences, social sciences, and arts and humanities, with searchable cited references. | Search by instrument name and sort results by times cited; view original articles describing the instrument development for clues on how to obtain Search by keyword and limit Run Cited Reference Search on the original article describing the instrument to find citing articles that may discuss validity and/or reliability of the instrument; OR search by instrument name in the Title field and additional terms such as validity, validation, reliability, etc. in the Topic field | |
- Last Updated: Apr 4, 2024 1:48 PM
- URL: https://researchguides.uvm.edu/researchinstruments
- Privacy Policy

Home » Questionnaire – Definition, Types, and Examples
Questionnaire – Definition, Types, and Examples
Table of Contents

Questionnaire
Definition:
A Questionnaire is a research tool or survey instrument that consists of a set of questions or prompts designed to gather information from individuals or groups of people.
It is a standardized way of collecting data from a large number of people by asking them a series of questions related to a specific topic or research objective. The questions may be open-ended or closed-ended, and the responses can be quantitative or qualitative. Questionnaires are widely used in research, marketing, social sciences, healthcare, and many other fields to collect data and insights from a target population.
History of Questionnaire
The history of questionnaires can be traced back to the ancient Greeks, who used questionnaires as a means of assessing public opinion. However, the modern history of questionnaires began in the late 19th century with the rise of social surveys.
The first social survey was conducted in the United States in 1874 by Francis A. Walker, who used a questionnaire to collect data on labor conditions. In the early 20th century, questionnaires became a popular tool for conducting social research, particularly in the fields of sociology and psychology.
One of the most influential figures in the development of the questionnaire was the psychologist Raymond Cattell, who in the 1940s and 1950s developed the personality questionnaire, a standardized instrument for measuring personality traits. Cattell’s work helped establish the questionnaire as a key tool in personality research.
In the 1960s and 1970s, the use of questionnaires expanded into other fields, including market research, public opinion polling, and health surveys. With the rise of computer technology, questionnaires became easier and more cost-effective to administer, leading to their widespread use in research and business settings.
Today, questionnaires are used in a wide range of settings, including academic research, business, healthcare, and government. They continue to evolve as a research tool, with advances in computer technology and data analysis techniques making it easier to collect and analyze data from large numbers of participants.
Types of Questionnaire
Types of Questionnaires are as follows:
Structured Questionnaire
This type of questionnaire has a fixed format with predetermined questions that the respondent must answer. The questions are usually closed-ended, which means that the respondent must select a response from a list of options.
Unstructured Questionnaire
An unstructured questionnaire does not have a fixed format or predetermined questions. Instead, the interviewer or researcher can ask open-ended questions to the respondent and let them provide their own answers.
Open-ended Questionnaire
An open-ended questionnaire allows the respondent to answer the question in their own words, without any pre-determined response options. The questions usually start with phrases like “how,” “why,” or “what,” and encourage the respondent to provide more detailed and personalized answers.
Close-ended Questionnaire
In a closed-ended questionnaire, the respondent is given a set of predetermined response options to choose from. This type of questionnaire is easier to analyze and summarize, but may not provide as much insight into the respondent’s opinions or attitudes.
Mixed Questionnaire
A mixed questionnaire is a combination of open-ended and closed-ended questions. This type of questionnaire allows for more flexibility in terms of the questions that can be asked, and can provide both quantitative and qualitative data.
Pictorial Questionnaire:
In a pictorial questionnaire, instead of using words to ask questions, the questions are presented in the form of pictures, diagrams or images. This can be particularly useful for respondents who have low literacy skills, or for situations where language barriers exist. Pictorial questionnaires can also be useful in cross-cultural research where respondents may come from different language backgrounds.
Types of Questions in Questionnaire
The types of Questions in Questionnaire are as follows:
Multiple Choice Questions
These questions have several options for participants to choose from. They are useful for getting quantitative data and can be used to collect demographic information.
- a. Red b . Blue c. Green d . Yellow
Rating Scale Questions
These questions ask participants to rate something on a scale (e.g. from 1 to 10). They are useful for measuring attitudes and opinions.
- On a scale of 1 to 10, how likely are you to recommend this product to a friend?
Open-Ended Questions
These questions allow participants to answer in their own words and provide more in-depth and detailed responses. They are useful for getting qualitative data.
- What do you think are the biggest challenges facing your community?
Likert Scale Questions
These questions ask participants to rate how much they agree or disagree with a statement. They are useful for measuring attitudes and opinions.
How strongly do you agree or disagree with the following statement:
“I enjoy exercising regularly.”
- a . Strongly Agree
- c . Neither Agree nor Disagree
- d . Disagree
- e . Strongly Disagree
Demographic Questions
These questions ask about the participant’s personal information such as age, gender, ethnicity, education level, etc. They are useful for segmenting the data and analyzing results by demographic groups.
- What is your age?
Yes/No Questions
These questions only have two options: Yes or No. They are useful for getting simple, straightforward answers to a specific question.
Have you ever traveled outside of your home country?
Ranking Questions
These questions ask participants to rank several items in order of preference or importance. They are useful for measuring priorities or preferences.
Please rank the following factors in order of importance when choosing a restaurant:
- a. Quality of Food
- c. Ambiance
- d. Location
Matrix Questions
These questions present a matrix or grid of options that participants can choose from. They are useful for getting data on multiple variables at once.
| The product is easy to use | ||||
| The product meets my needs | ||||
| The product is affordable |
Dichotomous Questions
These questions present two options that are opposite or contradictory. They are useful for measuring binary or polarized attitudes.
Do you support the death penalty?
How to Make a Questionnaire
Step-by-Step Guide for Making a Questionnaire:
- Define your research objectives: Before you start creating questions, you need to define the purpose of your questionnaire and what you hope to achieve from the data you collect.
- Choose the appropriate question types: Based on your research objectives, choose the appropriate question types to collect the data you need. Refer to the types of questions mentioned earlier for guidance.
- Develop questions: Develop clear and concise questions that are easy for participants to understand. Avoid leading or biased questions that might influence the responses.
- Organize questions: Organize questions in a logical and coherent order, starting with demographic questions followed by general questions, and ending with specific or sensitive questions.
- Pilot the questionnaire : Test your questionnaire on a small group of participants to identify any flaws or issues with the questions or the format.
- Refine the questionnaire : Based on feedback from the pilot, refine and revise the questionnaire as necessary to ensure that it is valid and reliable.
- Distribute the questionnaire: Distribute the questionnaire to your target audience using a method that is appropriate for your research objectives, such as online surveys, email, or paper surveys.
- Collect and analyze data: Collect the completed questionnaires and analyze the data using appropriate statistical methods. Draw conclusions from the data and use them to inform decision-making or further research.
- Report findings: Present your findings in a clear and concise report, including a summary of the research objectives, methodology, key findings, and recommendations.
Questionnaire Administration Modes
There are several modes of questionnaire administration. The choice of mode depends on the research objectives, sample size, and available resources. Some common modes of administration include:
- Self-administered paper questionnaires: Participants complete the questionnaire on paper, either in person or by mail. This mode is relatively low cost and easy to administer, but it may result in lower response rates and greater potential for errors in data entry.
- Online questionnaires: Participants complete the questionnaire on a website or through email. This mode is convenient for both researchers and participants, as it allows for fast and easy data collection. However, it may be subject to issues such as low response rates, lack of internet access, and potential for fraudulent responses.
- Telephone surveys: Trained interviewers administer the questionnaire over the phone. This mode allows for a large sample size and can result in higher response rates, but it is also more expensive and time-consuming than other modes.
- Face-to-face interviews : Trained interviewers administer the questionnaire in person. This mode allows for a high degree of control over the survey environment and can result in higher response rates, but it is also more expensive and time-consuming than other modes.
- Mixed-mode surveys: Researchers use a combination of two or more modes to administer the questionnaire, such as using online questionnaires for initial screening and following up with telephone interviews for more detailed information. This mode can help overcome some of the limitations of individual modes, but it requires careful planning and coordination.
Example of Questionnaire
Title of the Survey: Customer Satisfaction Survey
Introduction:
We appreciate your business and would like to ensure that we are meeting your needs. Please take a few minutes to complete this survey so that we can better understand your experience with our products and services. Your feedback is important to us and will help us improve our offerings.
Instructions:
Please read each question carefully and select the response that best reflects your experience. If you have any additional comments or suggestions, please feel free to include them in the space provided at the end of the survey.
1. How satisfied are you with our product quality?
- Very satisfied
- Somewhat satisfied
- Somewhat dissatisfied
- Very dissatisfied
2. How satisfied are you with our customer service?
3. How satisfied are you with the price of our products?
4. How likely are you to recommend our products to others?
- Very likely
- Somewhat likely
- Somewhat unlikely
- Very unlikely
5. How easy was it to find the information you were looking for on our website?
- Somewhat easy
- Somewhat difficult
- Very difficult
6. How satisfied are you with the overall experience of using our products and services?
7. Is there anything that you would like to see us improve upon or change in the future?
…………………………………………………………………………………………………………………………..
Conclusion:
Thank you for taking the time to complete this survey. Your feedback is valuable to us and will help us improve our products and services. If you have any further comments or concerns, please do not hesitate to contact us.
Applications of Questionnaire
Some common applications of questionnaires include:
- Research : Questionnaires are commonly used in research to gather information from participants about their attitudes, opinions, behaviors, and experiences. This information can then be analyzed and used to draw conclusions and make inferences.
- Healthcare : In healthcare, questionnaires can be used to gather information about patients’ medical history, symptoms, and lifestyle habits. This information can help healthcare professionals diagnose and treat medical conditions more effectively.
- Marketing : Questionnaires are commonly used in marketing to gather information about consumers’ preferences, buying habits, and opinions on products and services. This information can help businesses develop and market products more effectively.
- Human Resources: Questionnaires are used in human resources to gather information from job applicants, employees, and managers about job satisfaction, performance, and workplace culture. This information can help organizations improve their hiring practices, employee retention, and organizational culture.
- Education : Questionnaires are used in education to gather information from students, teachers, and parents about their perceptions of the educational experience. This information can help educators identify areas for improvement and develop more effective teaching strategies.
Purpose of Questionnaire
Some common purposes of questionnaires include:
- To collect information on attitudes, opinions, and beliefs: Questionnaires can be used to gather information on people’s attitudes, opinions, and beliefs on a particular topic. For example, a questionnaire can be used to gather information on people’s opinions about a particular political issue.
- To collect demographic information: Questionnaires can be used to collect demographic information such as age, gender, income, education level, and occupation. This information can be used to analyze trends and patterns in the data.
- To measure behaviors or experiences: Questionnaires can be used to gather information on behaviors or experiences such as health-related behaviors or experiences, job satisfaction, or customer satisfaction.
- To evaluate programs or interventions: Questionnaires can be used to evaluate the effectiveness of programs or interventions by gathering information on participants’ experiences, opinions, and behaviors.
- To gather information for research: Questionnaires can be used to gather data for research purposes on a variety of topics.
When to use Questionnaire
Here are some situations when questionnaires might be used:
- When you want to collect data from a large number of people: Questionnaires are useful when you want to collect data from a large number of people. They can be distributed to a wide audience and can be completed at the respondent’s convenience.
- When you want to collect data on specific topics: Questionnaires are useful when you want to collect data on specific topics or research questions. They can be designed to ask specific questions and can be used to gather quantitative data that can be analyzed statistically.
- When you want to compare responses across groups: Questionnaires are useful when you want to compare responses across different groups of people. For example, you might want to compare responses from men and women, or from people of different ages or educational backgrounds.
- When you want to collect data anonymously: Questionnaires can be useful when you want to collect data anonymously. Respondents can complete the questionnaire without fear of judgment or repercussions, which can lead to more honest and accurate responses.
- When you want to save time and resources: Questionnaires can be more efficient and cost-effective than other methods of data collection such as interviews or focus groups. They can be completed quickly and easily, and can be analyzed using software to save time and resources.
Characteristics of Questionnaire
Here are some of the characteristics of questionnaires:
- Standardization : Questionnaires are standardized tools that ask the same questions in the same order to all respondents. This ensures that all respondents are answering the same questions and that the responses can be compared and analyzed.
- Objectivity : Questionnaires are designed to be objective, meaning that they do not contain leading questions or bias that could influence the respondent’s answers.
- Predefined responses: Questionnaires typically provide predefined response options for the respondents to choose from, which helps to standardize the responses and make them easier to analyze.
- Quantitative data: Questionnaires are designed to collect quantitative data, meaning that they provide numerical or categorical data that can be analyzed using statistical methods.
- Convenience : Questionnaires are convenient for both the researcher and the respondents. They can be distributed and completed at the respondent’s convenience and can be easily administered to a large number of people.
- Anonymity : Questionnaires can be anonymous, which can encourage respondents to answer more honestly and provide more accurate data.
- Reliability : Questionnaires are designed to be reliable, meaning that they produce consistent results when administered multiple times to the same group of people.
- Validity : Questionnaires are designed to be valid, meaning that they measure what they are intended to measure and are not influenced by other factors.
Advantage of Questionnaire
Some Advantage of Questionnaire are as follows:
- Standardization: Questionnaires allow researchers to ask the same questions to all participants in a standardized manner. This helps ensure consistency in the data collected and eliminates potential bias that might arise if questions were asked differently to different participants.
- Efficiency: Questionnaires can be administered to a large number of people at once, making them an efficient way to collect data from a large sample.
- Anonymity: Participants can remain anonymous when completing a questionnaire, which may make them more likely to answer honestly and openly.
- Cost-effective: Questionnaires can be relatively inexpensive to administer compared to other research methods, such as interviews or focus groups.
- Objectivity: Because questionnaires are typically designed to collect quantitative data, they can be analyzed objectively without the influence of the researcher’s subjective interpretation.
- Flexibility: Questionnaires can be adapted to a wide range of research questions and can be used in various settings, including online surveys, mail surveys, or in-person interviews.
Limitations of Questionnaire
Limitations of Questionnaire are as follows:
- Limited depth: Questionnaires are typically designed to collect quantitative data, which may not provide a complete understanding of the topic being studied. Questionnaires may miss important details and nuances that could be captured through other research methods, such as interviews or observations.
- R esponse bias: Participants may not always answer questions truthfully or accurately, either because they do not remember or because they want to present themselves in a particular way. This can lead to response bias, which can affect the validity and reliability of the data collected.
- Limited flexibility: While questionnaires can be adapted to a wide range of research questions, they may not be suitable for all types of research. For example, they may not be appropriate for studying complex phenomena or for exploring participants’ experiences and perceptions in-depth.
- Limited context: Questionnaires typically do not provide a rich contextual understanding of the topic being studied. They may not capture the broader social, cultural, or historical factors that may influence participants’ responses.
- Limited control : Researchers may not have control over how participants complete the questionnaire, which can lead to variations in response quality or consistency.
About the author
Muhammad Hassan
Researcher, Academic Writer, Web developer
You may also like


Explanatory Research – Types, Methods, Guide

Textual Analysis – Types, Examples and Guide

Quantitative Research – Methods, Types and...

Ethnographic Research -Types, Methods and Guide

Descriptive Research Design – Types, Methods and...

Research Methods – Types, Examples and Guide

Research Instruments
- Resources for Identifying Instruments
- Assessing Instruments
- Obtaining the Full Instrument
- Getting Help
What are Research Instruments?
A research instrument is a tool used to collect, measure, and analyze data related to your subject.
Research instruments can be tests , surveys , scales , questionnaires , or even checklists .
To assure the strength of your study, it is important to use previously validated instruments!
Getting Started
Already know the full name of the instrument you're looking for?
- Start here!
Finding a research instrument can be very time-consuming!
This process involves three concrete steps:

It is common that sources will not provide the full instrument, but they will provide a citation with the publisher. In some cases, you may have to contact the publisher to obtain the full text.
Research Tip : Talk to your departmental faculty. Many of them have expertise in working with research instruments and can help you with this process.
- Next: Identifying a Research Instrument >>
- Last Updated: Aug 27, 2023 9:34 AM
- URL: https://guides.library.duq.edu/researchinstruments

- Foundations
- Write Paper
Search form
- Experiments
- Anthropology
- Self-Esteem
- Social Anxiety
- Survey Guide >
Types of Survey
There are various types of surveys you can choose from. Basically, the types of surveys are broadly categorized into two: according to instrumentation and according to the span of time involved. The types of surveys according to instrumentation include the questionnaire and the interview. On the other hand, the types of surveys according to the span of time used to conduct the survey are comprised of cross-sectional surveys and longitudinal surveys.
This article is a part of the guide:
- Response Scales
- Example - Questionnaire
- Advantages and Disadvantages
- Surveys and Questionnaires - Guide
- Personal Interview
Browse Full Outline
- 1 Surveys and Questionnaires - Guide
- 2.1 Research and Surveys
- 2.2 Advantages and Disadvantages
- 2.3 Survey Design
- 2.4 Sampling
- 3.1 Defining Goals
- 4.1 Survey Layout
- 4.2 Types of Questions
- 4.3 Constructing Questions
- 4.4 Response Formats
- 4.5 Response Scales
- 5.1 Selecting Method
- 5.2 Personal Interview
- 5.3 Telephone
- 5.4.1 Preparing Online Surveys
- 5.4.2 Online Tools
- 5.5 Focus Group
- 5.6 Panel Study
- 6.1 Pilot Survey
- 6.2 Increasing Response Rates
- 7.1 Analysis and Data
- 7.2 Conclusion
- 7.3 Presenting the Results
- 8 Example - Questionnaire
- 9 Checklist

According to Instrumentation
In survey research, the instruments that are utilized can be either a questionnaire or an interview (either structured or unstructured).
1. Questionnaires
Typically, a questionnaire is a paper-and-pencil instrument that is administered to the respondents. The usual questions found in questionnaires are closed-ended questions, which are followed by response options. However, there are questionnaires that ask open-ended questions to explore the answers of the respondents.
Questionnaires have been developed over the years. Today, questionnaires are utilized in various survey methods , according to how they are given. These methods include the self-administered, the group-administered, and the household drop-off. Among the three, the self-administered survey method is often used by researchers nowadays. The self-administered questionnaires are widely known as the mail survey method. However, since the response rates related to mail surveys had gone low, questionnaires are now commonly administered online, as in the form of web surveys.
- Advantages: Ideal for asking closed-ended questions; effective for market or consumer research
- Disadvantages: Limit the researcher’s understanding of the respondent’s answers; requires budget for reproduction of survey questionnaires
2. Interviews
Between the two broad types of surveys, interviews are more personal and probing. Questionnaires do not provide the freedom to ask follow-up questions to explore the answers of the respondents, but interviews do.
An interview includes two persons - the researcher as the interviewer, and the respondent as the interviewee. There are several survey methods that utilize interviews. These are the personal or face-to-face interview, the phone interview , and more recently, the online interview .
- Advantages: Follow-up questions can be asked; provide better understanding of the answers of the respondents
- Disadvantages: Time-consuming; many target respondents have no public-listed phone numbers or no telephones at all

According to the Span of Time Involved
The span of time needed to complete the survey brings us to the two different types of surveys: cross-sectional and longitudinal.
1. Cross-Sectional Surveys
Collecting information from the respondents at a single period in time uses the cross-sectional type of survey. Cross-sectional surveys usually utilize questionnaires to ask about a particular topic at one point in time. For instance, a researcher conducted a cross-sectional survey asking teenagers’ views on cigarette smoking as of May 2010. Sometimes, cross-sectional surveys are used to identify the relationship between two variables , as in a comparative study. An example of this is administering a cross-sectional survey about the relationship of peer pressure and cigarette smoking among teenagers as of May 2010.
2. Longitudinal Surveys
When the researcher attempts to gather information over a period of time or from one point in time up to another, he is doing a longitudinal survey. The aim of longitudinal surveys is to collect data and examine the changes in the data gathered. Longitudinal surveys are used in cohort studies , panel studies and trend studies.
- Psychology 101
- Flags and Countries
- Capitals and Countries
Sarah Mae Sincero (Sep 21, 2012). Types of Survey. Retrieved Sep 19, 2024 from Explorable.com: https://explorable.com/types-of-survey
You Are Allowed To Copy The Text
The text in this article is licensed under the Creative Commons-License Attribution 4.0 International (CC BY 4.0) .
This means you're free to copy, share and adapt any parts (or all) of the text in the article, as long as you give appropriate credit and provide a link/reference to this page.
That is it. You don't need our permission to copy the article; just include a link/reference back to this page. You can use it freely (with some kind of link), and we're also okay with people reprinting in publications like books, blogs, newsletters, course-material, papers, wikipedia and presentations (with clear attribution).
Want to stay up to date? Follow us!
Save this course for later.
Don't have time for it all now? No problem, save it as a course and come back to it later.
Footer bottom
- Privacy Policy

- Subscribe to our RSS Feed
- Like us on Facebook
- Follow us on Twitter
Survey Research and Administration
Survey research is a method in which data is collected from a target population, called the sample, by personal interviews, online surveys, the telephone, or paper questionnaires. Some forms of survey research such as online surveys may be completed in an automated fashion. The professionals at Statistics Solutions provide survey administration help to master’s and doctoral candidates in the survey administration phase of their research. The choice of survey instrument(s) used to gather data for your thesis or dissertation is critical. If you are planning to create your own survey instrument and administer it online (e.g., SurveyMonkey, QuestionPro, PsychData or Zoomerang), Statistics Solutions can help you create the survey questions and any subscales so they can be easily analyzed and answer your research questions. Our consultants can then help you validate your instrument and expedite the IRB approval process by helping you avoid the typical university and committee pitfalls. If you are using an established instrument , our statistical consultants will help you understand the validity and reliability information and the statistical analysis appropriate for the instrument constructs. Our statistical consultants will then help you integrate this information into your dissertation.

Discover How We Assist to Edit Your Dissertation Chapters
Aligning theoretical framework, gathering articles, synthesizing gaps, articulating a clear methodology and data plan, and writing about the theoretical and practical implications of your research are part of our comprehensive dissertation editing services.
- Bring dissertation editing expertise to chapters 1-5 in timely manner.
- Track all changes, then work with you to bring about scholarly writing.
- Ongoing support to address committee feedback, reducing revisions.
Key Terms and Concepts: Survey instrument: The questionnaire or response item posed to a respondent is called a survey research instrument . The instrument may be a questionnaire or an interview; it depends on the survey research. Interviews and questionnaires: An interview uses face-to-face interaction, whereas a questionnaire uses the mail and other indirect methods of taking responses from a respondent. Response structure: In survey research, the response structure is the format of the item. Structures may be open-ended, close-ended, multi-response, dichotomous, a ranking system, or a variety of other formats. Survey error: In survey research, survey errors includes factors such as the selection of the wrong sample, the wrong coding in a questionnaire, a tabulating error, data processing errors, interviewer bias, researcher bias, and misinterpretation of data. Pretesting: Pretesting refers to all the essential steps involved in survey research before selecting the final sample. According to Converse and Presser (1986: 65), two pretests should be conducted before selecting the final sample . Analysis of non-response: In survey research, some respondents do not fill out the entire questionnaire. The unanswered questions in this case become the missing values . We should exclude those values during the analysis or we should fill those missing values by using missing value analysis.
Data Collection Methods: Face-to-face interview: In survey research, this is the most expensive but reliable method for data collection. In face-to-face interviews, most of the respondents give complete and accurate answers. This method is used when the research requires deep exploration of opinion. Mail Survey: This method uses the Internet or sends mail to the respondents. There is no bias on the part of the interviewer in this method, but there is no control over respondent interaction. Telephone: This method is a fast method of data collection in survey research. This method supports open-ended responses and moderate control over interviewer bias. Web survey: This is a less expensive method and it is also the fastest method of data collection. This method is appropriate when we need data from a large population or when we need international data. This method is more suitable when we need unscientific but quick responses.
Survey Design Considerations: Survey layout: For Internet surveys or mail surveys, the layout of the survey should be attractive and easy to use; for example, the survey should avoid multiple fonts, the response area should be on the right side, there should be a clear separation of questions, and the survey should be an attractive color. Survey length: In survey research, the length of the survey should be as long as needed within the constraint of the respondent’s attention span. The surveys need to have a minimum of three items for testing a particular hypothesis.
Item bias in survey research: Ambiguity: Questions should be specific. We should avoid questions that make the respondent uncomfortable in giving the answer to that particular question. Rank lists: Respondents should not be asked to rank more than four or five items. Beyond that, respondents may give an arbitrary ranking just to get past the item. Unfamiliar terms and jargon: In survey research, we should not use unfamiliar words. Respondents must be able to answer the questions easily, and they cannot do this if the survey uses unfamiliar words or jargon. Poor grammatical format: In survey research, weak grammatical format can introduce bias. We should avoid poor grammatical format. Hypothetical items: We should not include hypothetical items. Hypothetical items make it difficult for the respondent to answer that particular question. Language differences: Items must have the same meaning when the questionnaire is given to populations speaking different languages. Types of items: Model items are those that measure variables in the survey model. Filter items: In survey research, filter items are those items which eliminate the unqualified respondents during post processing. Cross-check items: In survey research, cross-check items are those items which are used for consistency with the respondent. For example, at one place one can ask for the age of the respondent, and at another place, one can ask the data for the respondent’s birth. This will yield consistency of data.
Survey Administration Help Resources
Diment, K., & Garrett-Jones, S. (2007). How demographic characteristics affect mode preference in a postal/web mixed-mode survey of Australian researchers. Social Science Computer Review, 25 (3), 410-417.
Ehrlich, H. J. (1969). Attitudes, behavior, and the intervening variables. American Sociologist, 4 (1), 29-34.
Göritz , A. S. (2006). Cash lotteries as incentives in online panels. Social Science Computer Review, 24 (4), 445-459. Göritz, A. S., & Wolff, H. -G. (2007). Lotteries as incentives in longitudinal web studies. Social Science Computer Review, 25 (1), 99-110.
Groves, R. M., Cialdini, R. B., & Couper, M. P. (1992). Understanding the decision to participate in a survey. Public Opinion Quarterly, 56 (4), 475-495.
Healey, B. (2007). Drop downs and scroll mice: The effect of response option format and input mechanism employed on data quality in web surveys. Social Science Computer Review, 25 (1), 111-128.
Lee, S. (2006). An evaluation of nonresponse and coverage errors in a prerecruited probability web panel survey. Social Science Computer Review, 24 (4), 460-475.
Related Pages:
- Directory of Survey Instruments
- Missing Values in Data
Questionnaire Method In Research
Saul McLeod, PhD
Editor-in-Chief for Simply Psychology
BSc (Hons) Psychology, MRes, PhD, University of Manchester
Saul McLeod, PhD., is a qualified psychology teacher with over 18 years of experience in further and higher education. He has been published in peer-reviewed journals, including the Journal of Clinical Psychology.
Learn about our Editorial Process
Olivia Guy-Evans, MSc
Associate Editor for Simply Psychology
BSc (Hons) Psychology, MSc Psychology of Education
Olivia Guy-Evans is a writer and associate editor for Simply Psychology. She has previously worked in healthcare and educational sectors.
On This Page:
A questionnaire is a research instrument consisting of a series of questions for the purpose of gathering information from respondents. Questionnaires can be thought of as a kind of written interview . They can be carried out face to face, by telephone, computer, or post.
Questionnaires provide a relatively cheap, quick, and efficient way of obtaining large amounts of information from a large sample of people.

Data can be collected relatively quickly because the researcher would not need to be present when completing the questionnaires. This is useful for large populations when interviews would be impractical.
However, a problem with questionnaires is that respondents may lie due to social desirability. Most people want to present a positive image of themselves, and may lie or bend the truth to look good, e.g., pupils exaggerate revision duration.
Questionnaires can effectively measure relatively large subjects’ behavior, attitudes, preferences, opinions, and intentions more cheaply and quickly than other methods.
Often, a questionnaire uses both open and closed questions to collect data. This is beneficial as it means both quantitative and qualitative data can be obtained.
Closed Questions
A closed-ended question requires a specific, limited response, often “yes” or “no” or a choice that fit into pre-decided categories.
Data that can be placed into a category is called nominal data. The category can be restricted to as few as two options, i.e., dichotomous (e.g., “yes” or “no,” “male” or “female”), or include quite complex lists of alternatives from which the respondent can choose (e.g., polytomous).
Closed questions can also provide ordinal data (which can be ranked). This often involves using a continuous rating scale to measure the strength of attitudes or emotions.
For example, strongly agree / agree / neutral / disagree / strongly disagree / unable to answer.
Closed questions have been used to research type A personality (e.g., Friedman & Rosenman, 1974) and also to assess life events that may cause stress (Holmes & Rahe, 1967) and attachment (Fraley, Waller, & Brennan, 2000).
- They can be economical. This means they can provide large amounts of research data for relatively low costs. Therefore, a large sample size can be obtained, which should represent the population from which a researcher can then generalize.
- The respondent provides information that can be easily converted into quantitative data (e.g., count the number of “yes” or “no” answers), allowing statistical analysis of the responses.
- The questions are standardized. All respondents are asked exactly the same questions in the same order. This means a questionnaire can be replicated easily to check for reliability . Therefore, a second researcher can use the questionnaire to confirm consistent results.
Limitations
- They lack detail. Because the responses are fixed, there is less scope for respondents to supply answers that reflect their true feelings on a topic.
Open Questions
Open questions allow for expansive, varied answers without preset options or limitations.
Open questions allow people to express what they think in their own words. Open-ended questions enable the respondent to answer in as much detail as they like in their own words. For example: “can you tell me how happy you feel right now?”
Open questions will work better if you want to gather more in-depth answers from your respondents. These give no pre-set answer options and instead, allow the respondents to put down exactly what they like in their own words.
Open questions are often used for complex questions that cannot be answered in a few simple categories but require more detail and discussion.
Lawrence Kohlberg presented his participants with moral dilemmas. One of the most famous concerns a character called Heinz, who is faced with the choice between watching his wife die of cancer or stealing the only drug that could help her.
Participants were asked whether Heinz should steal the drug or not and, more importantly, for their reasons why upholding or breaking the law is right.
- Rich qualitative data is obtained as open questions allow respondents to elaborate on their answers. This means the research can determine why a person holds a certain attitude .
- Time-consuming to collect the data. It takes longer for the respondent to complete open questions. This is a problem as a smaller sample size may be obtained.
- Time-consuming to analyze the data. It takes longer for the researcher to analyze qualitative data as they have to read the answers and try to put them into categories by coding, which is often subjective and difficult. However, Smith (1992) has devoted an entire book to the issues of thematic content analysis that includes 14 different scoring systems for open-ended questions.
- Not suitable for less educated respondents as open questions require superior writing skills and a better ability to express one’s feelings verbally.
Questionnaire Design
With some questionnaires suffering from a response rate as low as 5%, a questionnaire must be well designed.
There are several important factors in questionnaire design.
Pilot Study
Question order.
Questions should progress logically from the least sensitive to the most sensitive, from the factual and behavioral to the cognitive, and from the more general to the more specific.
The researcher should ensure that previous questions do not influence the answer to a question.
Question order effects
- Question order effects occur when responses to an earlier question affect responses to a later question in a survey. They can arise at different stages of the survey response process – interpretation, information retrieval, judgment/estimation, and reporting.
- Types of question order effects include: unconditional (subsequent answers affected by prior question topic), conditional (subsequent answers depend on the response to the prior question), and associational (correlation between two questions changes based on order).
- Question order effects have been found across different survey topics like social and political attitudes, health and safety studies, vignette research, etc. Effects may be moderated by respondent factors like age, education level, knowledge and attitudes about the topic.
- To minimize question order effects, recommendations include avoiding judgmental dependencies between questions, separating potentially reactive questions, randomizing questions, following good survey design principles, considering respondent characteristics, and intentionally examining question context and order.
Terminology
- There should be a minimum of technical jargon. Questions should be simple, to the point, and easy to understand. The language of a questionnaire should be appropriate to the vocabulary of the group of people being studied.
- Use statements that are interpreted in the same way by members of different subpopulations of the population of interest.
- For example, the researcher must change the language of questions to match the social background of the respondent’s age / educational level / social class/ethnicity, etc.
Presentation
Ethical issues.
- The researcher must ensure that the information provided by the respondent is kept confidential, e.g., name, address, etc.
- This means questionnaires are good for researching sensitive topics as respondents will be more honest when they cannot be identified.
- Keeping the questionnaire confidential should also reduce the likelihood of psychological harm, such as embarrassment.
- Participants must provide informed consent before completing the questionnaire and must be aware that they have the right to withdraw their information at any time during the survey/ study.
Problems with Postal Questionnaires
At first sight, the postal questionnaire seems to offer the opportunity to get around the problem of interview bias by reducing the personal involvement of the researcher. Its other practical advantages are that it is cheaper than face-to-face interviews and can quickly contact many respondents scattered over a wide area.
However, these advantages must be weighed against the practical problems of conducting research by post. A lack of involvement by the researcher means there is little control over the information-gathering process.
The data might not be valid (i.e., truthful) as we can never be sure that the questionnaire was completed by the person to whom it was addressed.
That, of course, assumes there is a reply in the first place, and one of the most intractable problems of mailed questionnaires is a low response rate. This diminishes the reliability of the data
Also, postal questionnaires may not represent the population they are studying. This may be because:
- Some questionnaires may be lost in the post, reducing the sample size.
- The questionnaire may be completed by someone not a member of the research population.
- Those with strong views on the questionnaire’s subject are more likely to complete it than those without interest.
Benefits of a Pilot Study
A pilot study is a practice / small-scale study conducted before the main study.
It allows the researcher to try out the study with a few participants so that adjustments can be made before the main study, saving time and money.
It is important to conduct a questionnaire pilot study for the following reasons:
- Check that respondents understand the terminology used in the questionnaire.
- Check that emotive questions are not used, as they make people defensive and could invalidate their answers.
- Check that leading questions have not been used as they could bias the respondent’s answer.
- Ensure the questionnaire can be completed in an appropriate time frame (i.e., it’s not too long).
Frequently Asked Questions
How do psychological researchers analyze the data collected from questionnaires.
Psychological researchers analyze questionnaire data by looking for patterns and trends in people’s responses. They use numbers and charts to summarize the information.
They calculate things like averages and percentages to see what most people think or feel. They also compare different groups to see if there are any differences between them.
By doing these analyses, researchers can understand how people think, feel, and behave. This helps them make conclusions and learn more about how our minds work.
Are questionnaires effective in gathering accurate data?
Yes, questionnaires can be effective in gathering accurate data. When designed well, with clear and understandable questions, they allow individuals to express their thoughts, opinions, and experiences.
However, the accuracy of the data depends on factors such as the honesty and accuracy of respondents’ answers, their understanding of the questions, and their willingness to provide accurate information. Researchers strive to create reliable and valid questionnaires to minimize biases and errors.
It’s important to remember that while questionnaires can provide valuable insights, they are just one tool among many used in psychological research.
Can questionnaires be used with diverse populations and cultural contexts?
Yes, questionnaires can be used with diverse populations and cultural contexts. Researchers take special care to ensure that questionnaires are culturally sensitive and appropriate for different groups.
This means adapting the language, examples, and concepts to match the cultural context. By doing so, questionnaires can capture the unique perspectives and experiences of individuals from various backgrounds.
This helps researchers gain a more comprehensive understanding of human behavior and ensures that everyone’s voice is heard and represented in psychological research.
Are questionnaires the only method used in psychological research?
No, questionnaires are not the only method used in psychological research. Psychologists use a variety of research methods, including interviews, observations , experiments , and psychological tests.
Each method has its strengths and limitations, and researchers choose the most appropriate method based on their research question and goals.
Questionnaires are valuable for gathering self-report data, but other methods allow researchers to directly observe behavior, study interactions, or manipulate variables to test hypotheses.
By using multiple methods, psychologists can gain a more comprehensive understanding of human behavior and mental processes.
What is a semantic differential scale?
The semantic differential scale is a questionnaire format used to gather data on individuals’ attitudes or perceptions. It’s commonly incorporated into larger surveys or questionnaires to assess subjective qualities or feelings about a specific topic, product, or concept by quantifying them on a scale between two bipolar adjectives.
It presents respondents with a pair of opposite adjectives (e.g., “happy” vs. “sad”) and asks them to mark their position on a scale between them, capturing the intensity of their feelings about a particular subject.
It quantifies subjective qualities, turning them into data that can be statistically analyzed.
Ayidiya, S. A., & McClendon, M. J. (1990). Response effects in mail surveys. Public Opinion Quarterly, 54 (2), 229–247. https://doi.org/10.1086/269200
Fraley, R. C., Waller, N. G., & Brennan, K. A. (2000). An item-response theory analysis of self-report measures of adult attachment. Journal of Personality and Social Psychology, 78, 350-365.
Friedman, M., & Rosenman, R. H. (1974). Type A behavior and your heart . New York: Knopf.
Gold, R. S., & Barclay, A. (2006). Order of question presentation and correlation between judgments of comparative and own risk. Psychological Reports, 99 (3), 794–798. https://doi.org/10.2466/PR0.99.3.794-798
Holmes, T. H., & Rahe, R. H. (1967). The social readjustment rating scale. Journal of psychosomatic research, 11(2) , 213-218.
Schwarz, N., & Hippler, H.-J. (1995). Subsequent questions may influence answers to preceding questions in mail surveys. Public Opinion Quarterly, 59 (1), 93–97. https://doi.org/10.1086/269460
Smith, C. P. (Ed.). (1992). Motivation and personality: Handbook of thematic content analysis . Cambridge University Press.
Further Information
- Questionnaire design and scale development
- Questionnaire Appraisal Form

Finding Research Instruments, Surveys, and Tests: Home
- Create Tests
- Search for Tests
- Online Test Sources
- Dissertations/Theses
- More about ERIC
- Citing in APA Style
- More about MMY
- More about PsycINFO
What are Research Instruments
A research instrument is a survey, questionnaire, test, scale, rating, or tool designed to measure the variable(s), characteristic(s), or information of interest, often a behavioral or psychological characteristic. Research instruments can be helpful tools to your research study.
"Careful planning for data collection can help with setting realistic goals. Data collection instrumentation, such as surveys, physiologic measures (blood pressure or temperature), or interview guides, must be identified and described. Using previously validated collection instruments can save time and increase the study's credibility. Once the data collection procedure has been determined, a time line for completion should be established." (Pierce, 2009, p. 159)
- Pierce, L.L. (2009). Twelve steps for success in the nursing research journey. Journal of Continuing Education in Nursing 40(4), 154-162.
A research instrument is developed as a method of data generation by researchers and information about the research instrument is shared in order to establish the credibility and validity of the method. Whether other researchers may use the research instrument is the decision of the original author-researchers. They may make it publicly available for free or for a price or they may not share it at all. Sources about research instruments have a purpose of describing the instrument to inform. Sources may or may not provide the instrument itself or the contact information of the author-researcher. The onus is on the reader-researcher to try to find the instrument itself or to contact the author-researcher to request permission for its use, if necessary.
How to choose the right one?
Are you trying to find background information about a research instrument? Or are you trying to find and obtain an actual copy of the instrument?
If you need information about a research instrument, what kind of information do you need? Do you need information on the structure of the instrument, its content, its development, its psychometric reliability or validity? What do you need?
If you plan to obtain an actual copy of the instrument to use in research, you need to be concerned not only with obtaining the instrument, but also obtaining permission to use the instrument. Research instruments may be copyrighted. To obtain permission, contact the copyright holder in writing (print or email).
If someone posts a published test or instrument without the permission of the copyright holder, they may be violating copyright and could be legally liable.
What are you trying to measure? For example, if you are studying depression, are you trying to measure the duration of depression, the intensity of depression, the change over time of the episodes, … what? The instrument must measure what you need or it is useless to you.
Factors to consider when selecting an instrument are • Well-tested factorial structure, validity & reliability • Availability of supportive materials and technology for entering, analyzing and interpreting results • Availability of normative data as a reference for evaluating, interpreting, or placing in context individual test scores • Applicable to wide range of participants • Can also be used as personal development tool/exercise • User-friendliness & administrative ease • Availability; can you obtain it? • Does it require permission from the owner to use it? • Financial cost • Amount of time required
Check the validity and reliability of tests and instruments. Do they really measure what they claim to measure? Do they measure consistently over time, with different research subjects and ethnic groups, and after repeated use? Research articles that used the test will often include reliability and validity data.
How Locate Instrument
Realize that searching for an instrument may take a lot of time. They may be published in a book or article on a particular subject. They be published and described in a dissertation. They may posted on the Internet and freely available. A specific instrument may be found in multiple publications and have been used for a long time. Or it may be new and only described in a few places. It may only be available by contacting the person who developed it, who may or may not respond to your inquiry in a timely manner.
There are a variety of sources that may used to search for research instruments. They include books, databases, Internet search engines, Web sites, journal articles, and dissertations.
A few key sources and search tips are listed in this guide.
Permission to Use the Test
If you plan to obtain an actual copy of the instrument to use in research, you need to be concerned not only with obtaining the instrument, but also obtaining permission to use the instrument. Research instruments are copyrighted. To obtain permission, contact the copyright holder to obtain permission in writing (print or email). Written permission is a record that you obtained permission.
It is a good idea to have them state in wiritng that they are indeed the copyright holder and that they grant you permission to use the instrument. If you wish to publish the actual instrument in your paper, get permission for that, too. You may write about the instrument without obtaining permission. (But remember to cite it!)
If someone posts a published test or instrument without the permission of the copyright holder, they are violating copyright and could be legally liable.
Subject Guide

- Next: Create Tests >>
- Last Updated: Jul 30, 2024 9:45 AM
- URL: https://library.indianastate.edu/instruments
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
The PMC website is updating on October 15, 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Sage Choice

Designing and Using Surveys in Nursing Research: A Contemporary Discussion
Siobhan o’connor.
1 National University of Ireland Galway, Ireland
This commentary summarizes the contemporary design and use of surveys or questionnaires in nursing science, particularly in light of recent reporting guidelines to standardize and improve the quality of survey studies in healthcare research. The benefits, risks, and limitations of these types of data collection tools are also briefly discussed.
The use of research questionnaires or surveys in nursing is a long standing tradition, dating back to the 1960s ( Logan, 1966 ) and 1970s ( Oberst, 1978 ), when the scientific discipline emerged. This type of tool enables nursing researchers to gather primary data from a specific population, whether it is patients, carers, nurses, or other stakeholders to address gaps in the existing evidence base of a particular clinical, pedagogical, or policy area. However, the recent creation of a checklist for reporting survey studies called CROSS: Consensus-Based Checklist for Reporting of Survey Studies, hints at problems in their design, development, administration, and reporting ( Sharma et al., 2021 ). This commentary discussion focuses on the process of developing, validating, and administering surveys in nursing research and some ways to strengthen this methodological approach.
Ideally, surveys should be constructed to gather the minimum amount of information from respondents to provide good quality data about a problem or phenomenon. Gathering large amounts of unnecessary data may complicate a survey, leading to a low response rate and weak findings. Therefore, time and expertise is needed when designing research surveys ( Robb & Shellenbarger, 2020 ). Firstly, existing evidence should be reviewed to identify if an existing survey could be utilized or refined. The COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN), and associated database and critical appraisal checklist, could be employed to examine the psychometric properties of an established tool and its methodological quality before use ( Mokkink et al., 2016 ). For instance, Charette et al. (2020) followed the COSMIN approach when conducting a systematic review of the psychometric properties of scales that assessed new nurses clinical competence.
If a new instrument needs to be developed, then reviewing relevant literature could help inform what should be measured for example, what people know, think, feel, or do, along with guiding the content of specific survey questions ( Polit & Beck, 2020 ). Other techniques can be employed to create questions including a Delphi study to gather expert opinion ( Bender et al., 2018 ), or focus groups with patients, clinicians, educators, students, or policy makers ( Tajik et al., 2010 ). Decisions about the style of survey questions also needs consideration as each brings advantages and disadvantages. An open question gives a respondent free reign with their answer which could uncover fresh insights on a topic. However, it may contribute to respondent fatigue if too many are asked, and the data can be time consuming to analyze ( O’Cathain & Thomas, 2004 ).
How each question is worded is also important to avoid leading, composite, or presumptive questions, ones that are vague, overly lengthy and complex, or include double negatives, jargon, or terminology unfamiliar to the reader, so that what is being asked and answered is clear and consistent. The sequence of questions should also be logical, opening with more general non-threatening questions, followed by more specific ones that can be grouped or filtered accordingly, and closing with socio-demographic variables and a thank you ( Boynton & Greenhalgh, 2004 ). Closed fixed choice questions can be formulated in a number of ways including checklists, frequency or Likert-type scales, Guttman or cumulative scales, Thurstone scales, and rankings, which vary in their content, structure, and layout, and require either a dichotomous or multiple-choice response. The sensitivity of any measurement scale is important to ensure it accurately represents the respondents answer and reduces the risk of bias ( Polit, 2014 ). Hence, piloting a draft survey with a small sample of intended respondents can help identify problems with ambiguous content, the format of questions, or confusing instructions or layouts.
The validity and reliability of a survey instrument should also be established to demonstrate the questions are worded appropriately and illicit accurate answers. Validity is about accuracy, in terms of how well a survey measures what it is supposed to. It can be assessed in three ways: (1) face or content validity, (2) construct validity, and (3) criterion validity ( Rattray & Jones, 2007 ). Content validity looks at comprehensiveness, and whether questions adequately measure all aspects of a subject matter. For example, Devriendt et al. (2012) examined the content validity of the Safety Attitudes Questionnaire through expert clinician review and using the content validity index and a modified kappa index. Construct validity focuses on whether the concept(s) that underpin the questions in a survey correspond with contemporary theory and scientific knowledge. For instance, McSherry et al. (2002) employed factor analysis to determine construct validity for a Spirituality and Spiritual Care Rating Scale. Numerous research studies are often required to evaluate and refine the construct validity of a survey instrument to ensure it is robust. Some go further and investigate both convergent and discriminate validity, the two sub-types of construct validity ( Hallett et al., 2018 ; Zhao et al., 2020 )
Criterion validity refers to how much the scores in a survey measure agree with a gold-standard that is considered an ideal measure of the constructs or variables. This approach is not always feasible, if there are no reliable measures for independent comparison ( Polit & Beck, 2020 ). It can be done in two ways, the first by calculating a correlation coefficient which tests the strength of the association (not agreement) between the results from a survey and an external independent measure. Secondly, sensitivity and specificity can be calculated, although there is usually a trade-off between the two ( Groves, 2009 ). Sensitivity measures the ability of a survey to detect all instances of its subject matter (true positives), while specificity measures the ability of a survey to discriminate all instances of its subject matter from those which are not related (true negatives). Both false negatives and false positive errors may occur, so the nature of the research and survey instrument should guide which type of error should be minimized as much as possible ( Dillman, 2014 ). The two types of criterion validity, concurrent and predictive validity, can also be measured. Concurrent validity compares survey questions or items to a related validated measure, both of which are assessed at the same time, whereas predictive validity compares survey items against some criterion measure at a later time ( Kim & Abraham, 2016 ). While validity testing can be time consuming, expensive, and require a significant amount of statistical expertise, it is a robust way to develop and improve the psychometric properties of surveys.
The other major concept used to evaluate the quality of surveys is reliability which focuses on the consistency of a survey and its items, to ensure it would give the same results if repeated under the same conditions ( Rattray & Jones, 2007 ). The three kinds of reliability testing are: (1) test retest, (2) inter-rater, and (3) internal consistency. Test retest looks at consistency of a measure across time and whether survey results from the same person were the same on at least two or more occasions. This can be measured using a number of statistical techniques such as the intraclass correlation coefficient and Wilcoxon signed rank test ( Lovén Wickman et al., 2019 ). Inter-rater reliability examines the consistency of a measure across raters or observers to determine if a person scores items in a survey in the same way multiple times. Cohen’s kappa ( Dancey et al., 2012 ) and the intra-class correlation coefficient ( Ryu et al., 2013 ) are common statistical measures for this. Finally, internal consistency is how consistently respondents’ answer items in a survey, if pairs of questions measuring the same concept are asked in different ways which can be calculated using Chronbach’s alpha ( Paans et al., 2010 ). Although reliability may be established and the survey results reproducible, this does not mean they are valid and may be incorrect unless the instruments’ validity is also determined.
Once a survey is designed, it then needs to be administered to the appropriate population, once ethical approval is granted. Self-completing surveys are the most common as they can quickly and easily be given to a large population using online, electronic or paper methods which are affordable options. An interview administered survey is an alternative, where questions are answered in the presence of a researcher, if sensitive topics need to be discussed, if vulnerable populations need to be reached, or if a survey is long and complex. A Cochrane review of interventions for administering postal and electronic questionnaires reported several strategies such as utilizing stamped addressed envelopes, financial incentives, and personalized communications, were effective in increasing the response rate ( Edwards et al., 2009 ), as low response rates can negatively impact the results of survey studies. After data is gathered, verified, cleaned, and anonymized, it needs to be coded using suitable analyses. Epi Info™ ( https://www.cdc.gov/epiinfo/index.html ) is a popular tool for entering and storing survey data before exporting it to a statistical analysis package ( Kebede et al., 2017 ).
Finally, surveys are frequently published in scientific nursing journals. However, Sharma et al. (2021) highlighted the substantial variability and inconsistency in how research surveys are reported which can weaken the evidence base on a topic. They emphasized that despite two guidelines for reporting non-web and web-based surveys, SURGE ( Grimshaw, 2014 ) and CHERRIES ( Eysenbach, 2004 ), improvements in the reporting of survey research have not materialized and these tools have limitations. Hence, a new comprehensive survey reporting guideline called CROSS was developed to enhance the transparency and replicability of survey based health research ( Sharma et al., 2021 ). This new guideline should be used in nursing to enable survey studies to be better designed, conducted, and reported. By undertaking rigorous, high-quality surveys, researchers can advance nursing science, strengthen the evidence base on which nurses practice, and help make a positive impact on patient care and health service delivery.
Author Biography
Siobhan O’Connor , BSc, CIMA CBA, BSc, RGN, PhD, is a Lecturer at the School of Nursing and Midwifery, National University of Ireland Galway, Ireland. She has a multidisciplinary background in both nursing and information systems. Hence, her research focuses on the design, implementation, and use of a range of technologies in healthcare.
Author Contributions: The sole author drafted and wrote the manuscript.
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.

A .gov website belongs to an official government organization in the United States.
A lock ( ) or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.
- Surveys and Vitals Systems
- Budget and Funding
- Data and Analysis Tools
- Partnerships
- Board of Scientific Counselors
- Policies and Procedures
- Employment at NCHS
- Responses to Health Data Requests
- Email Updates
Related Topics:
- View All Home
- NCHS Publications
- NCHS Pressroom
SANDS—Semi-Automated Non-response Detection for Surveys
- When analyzing data, researchers sometimes need to filter out responses that may not or do not answer the question.
- The SANDS—Semi-Automated Non-response Detection for Surveys—model helps researchers process survey response data by detecting these non-responses.
- Learn more about how SANDS helps researchers review large amounts of survey response data.
About SANDS
SANDS—Semi-Automated Non-response Detection for Surveys—is an open-access AI tool developed by the National Center for Health Statistics (NCHS). It helps researchers and survey administrators detect responses that may not or do not answer the question (non-responses) in open-ended survey text. The model helps human reviewers to quickly divide a large volume of text for manual review.
To use SANDS, follow the model card or the detailed instructions in the Getting Started section.
Before applying the model to real data, review these sections:
- Misuses and Out-of-scope
- Risks, Limitations, and Biases
Model details
This model is a fine-tuned version of the supervised SimCSE BERT base uncased model . It was introduced at the American Association of Public Opinion Research (AAPOR) 2022 Annual Conference .
The model is uncased, so it treats important , Important , and ImPoRtAnT the same.
- Developed by: CDC's National Center for Health Statistics
- Model Type: Text Classification
- Language(s): English
- License: Apache-2.0
Parent Model: For more details about SimCSE, we encourage users to visit the SimCSE Github repository , and the base model on HuggingFace. The model was fine-tuned on 3,000 labeled, open-ended responses from the NCHS Research and Development Survey's RANDS during COVID 19 Rounds 1 and 2 surveys. The base SimCSE BERT model was trained on BookCorpus and English Wikipedia.
Training procedure
- Learning rate: 5e-5
- Batch size: 16
- Number training epochs: 4
- Base Model pooling dimension: 768
- Number of labels: 5
Getting started
To use SANDS, first install python. Using a package manager, install pandas and the transformers module:
> pip install transformers pandas
Once you’ve installed the modules, the following code illustrates how to download the model, and parse a fixed set of responses:
from transformers import AutoTokenizer, AutoModelForSequenceClassification import torch import pandas as pd
# Load the model model_location = "NCHS/SANDS" model = AutoModelForSequenceClassification.from_pretrained(model_location) tokenizer = AutoTokenizer.from_pretrained(model_location)
# Create example responses to test responses = [ "sdfsdfa", "idkkkkk", "Because you asked", "I am a cucumber", "My job went remote and I needed to take care of my kids", ]
# Run the model and compute a score for each response with torch.no_grad(): tokens = tokenizer(responses, padding=True, truncation=True, return_tensors="pt") output = model(**tokens) scores = torch.softmax(output.logits, dim=1).numpy()
# Display the scores in a table columns = ["Gibberish", "Uncertainty", "Refusal", "High-risk", "Valid"] df = pd.DataFrame(scores, columns=columns) df.index.name = "Response" print(df)
The code should output the following table
| sdfsdfa | 0.998 | 0.000 | 0.000 | 0.000 | 0.000 |
| idkkkkk | 0.002 | 0.995 | 0.001 | 0.001 | 0.001 |
| Because you asked | 0.001 | 0.001 | 0.976 | 0.006 | 0.014 |
| I am a cucumber | 0.001 | 0.001 | 0.002 | 0.797 | 0.178 |
| My job went remote and I needed to take care of my kids | 0.000 | 0.000 | 0.000 | 0.000 | 1.000 |
This model is to be used on survey responses for data cleaning. When analyzing data, researchers can use SANDS to filter out non-responses. The model will return a score for a response in 5 different categories:
- Uncertainty
- Valid as a probability vector that sums to 1
Response types
- Examples: "ksdhfkshgk" and "sadsadsadsadsadsadsad"
- Examples: "Because" or "Meow"
- Examples: "I dont know" or "unsure what you are asking"
- Examples: "Necessity" or "I am a cucumber"
- Examples: "COVID began for me when my children's school went online and I needed to stay home to watch them" or "staying home, avoiding crowds, still wear masks"
Misuses and out-of-scope use
The model has been trained εspecifically to identify survey non-response when the survey respondent has given an open-ended response, but their answer does not address the question or provide meaningful insight. Examples of these types of responses include "meow," "ksdhfkshgk," or "idk."
The model was fine-tuned on 3,000 labeled, open-ended responses to web probes on questions relating to the COVID-19 pandemic. These responses were gathered from NCHS's Research and Development Survey .
Web probes are questions designed to draw out information about how respondents understand, think about, and respond to the questions that are being evaluated. They are different than traditional open-ended survey questions. The context of our labeled responses was limited in focus to both COVID-19 and health responses. Responses outside this scope may notice a drop in performance.
The model trained on responses from both web and phone-based open-ended probes. There may be limitations in model effectiveness with more traditional open-ended survey questions with responses provided in other mediums.
This model does not assess the factual accuracy of responses or filter out responses with different demographic biases. It was not trained to be factual of people or events, so using the model for such classification is out of scope for the abilities of the model.
We did not train the model to recognize non-response in any language other than English. Responses in languages other than English are out of scope and the model will perform poorly. Any correct classifications are a result of the base SimCSE or Bert Models.
Risk, limitations, and biases
To investigate if there were differences between demographic groups on sensitivity and specificity, we conducted two-tailed Z-tests across demographic groups. These included:
- Education (some college or less and bachelor’s or more)
- Sex (male or female)
- Mode (computer or telephone)
- Race and ethnicity (non-Hispanic White, non-Hispanic Black, Hispanic, and all others who are non-Hispanic)
- Age (18-29, 30-44, 45-59, and 60+)
There were 4,813 responses to 3 probes. To control for family-wise error rate, we applied the Bonferroni correction to the alpha level (α < 0.00167).
There were statistically significant differences in specificity between education levels, mode, and White and Black respondents. There were no statistically significant differences in sensitivity.
Respondents with some college or less had lower specificity compared to those with more education (0.73 versus 0.80, p < 0.0001). Respondents who used a smartphone or computer to complete their survey had a higher specificity than those who completed the survey over the telephone (0.77 versus 0.70, p < 0.0001). Black respondents had a lower specificity than White respondents (0.65 versus 0.78, p < 0.0001). Effect sizes for education and mode were small (h = 0.17 and h = 0.16, respectively). The effect size for race was between small and medium (h = 0.28).
Because the model was fine-tuned from SimCSE, itself fine-tuned from BERT, it will reproduce all biases inherent in these base models. Due to tokenization, the model may incorrectly classify typos, especially in acronyms. For example: LGBTQ is valid, while LBGTQ is classified as gibberish.
Open source license
Model and code are released as open source under the Creative Commons Universal Public Domain. That includes source files and code samples, if any, in the content. This means you can use the code, model, and content in this repository except for any official trademarks in your own projects.
Open-source projects are made available and contributed to under licenses that include terms that—for the protection of contributors—make clear that the projects are offered—
- “As-is”
- Without warranty
- Disclaiming liability for damages resulting from using the projects
This model is no different. The open content license it is offered under includes such terms.
National Center for Health Statistics
Homepage for the National Center for Health Statistics, the nation's provider of official health statistics
- Open access
- Published: 18 September 2024
Structural validation of two person-centred practice inventories PCPI-S and PCPI-C - French version
- Cedric Mabire 1 ,
- Marie Piccot-Crezollet 2 ,
- Vaibhav Tyagi 3 ,
- Brendan McCormack 3 &
- Joanie Pellet 1
BMC Health Services Research volume 24 , Article number: 1092 ( 2024 ) Cite this article
Metrics details
The shift towards person-centred care has become integral in achieving high-quality healthcare, focusing on individual patient needs, preferences, and values. However, existing instruments for measuring person-centred practice often lack theoretical underpinnings and comprehensive assessment. The Person-centred Practice Inventory – Staff (PCPI-S) and the Person-centred Practice Inventory – Care (PCPI-C) were developed in English to measure clinicians’ and patients’ experience of person-centred practice. The aim of this study was to investigate the psychometric properties of the French version of the PCPI-S and PCPI-C.
A multi-centred cross-sectional study was conducted in six hospitals in French-speaking Switzerland. Construct validity of the PCPI-S and the PCPI-C was evaluated by using confirmatory factor analysis and McDonald’s Omega coefficient was used to determine the internal consistency.
A sample of 558 healthcare professionals and 510 patients participated in the surveys. Psychometric analyses revealed positive item scores and acceptable factor loadings, demonstrating the meaningful contribution of each item to the measurement model. The Omega coefficient indicated acceptable to excellent internal consistency for the constructs. Model fit statistics demonstrated good model fit for the PCPI-S and PCPI-C.
Conclusions
The findings support the construct validity and internal consistency of the PCPI-S and PCPI-C in assessing person-centred practice among healthcare professionals and patients in French-speaking Switzerland. This validation offers valuable tools for evaluating person-centred care in hospital settings.
Peer Review reports
Person-centred care is an approach to healthcare that prioritises the individual needs, preferences, and values of the patient [ 1 ]. This approach recognises the fundamental role of patients as active participants in their own care, emphasizes the genuine relationship between patients and health professionals and acknowledges the context in which the care is delivered [ 2 ]. The shift towards person-centred care has gained momentum over the past few decades and become essential for achieving high-quality healthcare [ 3 ]. Person-centred care is of particular interest to politicians, researchers, and clinicians, as it is associated with improved clinical outcomes [ 4 , 5 ], patient satisfaction [ 4 , 6 , 7 ], work environment factors [ 8 ] and economic outcomes [ 9 , 10 ]. Person-centred care has been implemented across various healthcare settings, including primary care, long-term care and acute care facilities [ 11 , 12 ].
The Person-centred Practice Framework (PCPF) was developed by McCormack and McCance to support healthcare professionals to understand the dimensions of person-centredness and how to implement person-centred care in clinical practice. The PCP Framework comprises five interrelated domains: macro-context, prerequisites, care environment, person‐centred processes, and person‐centred outcomes. The macro-context domain refers to broader societal, cultural, and policy-related factors that influence healthcare practices. The prerequisites domain emphasises the essential organisational and practice-level elements required to support person-centred care. The care environment domain centres on the physical and emotional context in which care is provided. The person-centred processes domain highlights the importance of effective communication, engagement, and collaborative decision-making between patients and healthcare providers, fostering meaningful partnerships in care. Finally, the person-centred outcomes domain focuses on the positive impacts of person-centred care on patients [ 1 ].
Evaluation of person-centred practice is essential for identifying areas for improvement and monitoring its effective implementation within healthcare organisations [ 13 ]. Measurement tools can provide a standardised approach to assess the extent to which care aligns with person-centred principles and to support healthcare professionals in enhancing quality-of-care delivery and tailoring services to meet individual needs [ 14 ]. However, most of the available instruments measuring person-centred practice lack theoretical underpinnings or fail to assess the various aspects of person-centred care comprehensively [ 14 , 15 ]. To address the need of demonstrating the value of person-centred care, the PCPF has guided the development of measurement tools. The Person-Centred Practice Inventory – Staff (PCPI-S) developed by Slater et al. and the Person-Centred Practice Inventory – Care (PCPI-C) are aligned with key dimensions of the PCP Framework, including prerequisites, care environment, and person-centred processes [ 1 , 16 , 17 ]. The psychometric properties of the original version of the PCPI-S are acceptable (root mean square error of approximation (RMSEA) = 0.053, comparative fit index (CFI) = 0.951) with reference to the COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) criteria: CFI > 0.95, RMSEA < 0.06, standardised root mean residual (SRMR) < 0.08 [ 16 , 18 ]. The PCPI-S was designed for and tested with health care staff across all healthcare settings [ 16 , 19 – 24 ]. The instrument has been developed in English [ 9 ] and then translated into Swiss German, German, Austrian, Norwegian, Malaysian, Spanish and Portuguese [ 19 – 25 ]. The psychometric properties of the original version of the PCPI-C have not yet been published. By capturing the perspectives of both healthcare professionals and patients, the PCPI-S and the PCPI-C provide a comprehensive assessment of person-centred care [ 16 ]. Validation efforts are required to determine whether the PCPI-S and the PCPI-C translated into French provide valid measures of person-centred practice [ 16 ].
The aim of this study was to evaluate the construct validity and internal consistency of the PCPI-S and the PCPI-C among health care staff and patients in the French-speaking part of Switzerland.
Design and setting
This multi-centred cross-sectional study was conducted between March and August 2022. We invited Chief Nursing Officers (CNOs) of major public hospitals in the French-speaking part of Switzerland to participate. Out of those contacted, six hospitals agreed to take part in the study. Following this initial outreach, the project was introduced to the departments selected by the CNO. Subsequently, the unit participation was determined by the management teams. Notably, there were no specific criteria for the selection of units, as the PCPI-S and PCPI-C were intended for use across various healthcare settings and by professionals of different disciplines. Participating study sites included medical and surgical units, obstetrics/gynaecology/maternity, oncology, rehabilitation and geriatrics, neurology, outpatient care, and psychiatry.
- Person-centred practice inventory
The PCPI-S and the PCPI-C were translated into French prior to this study by using principles of good practice for the translation and cultural adaptation of patient reported outcomes measures [ 26 ]. Two nurses with a master’s degree, independently translated the PCPI-S and PCPI-C into French and then confer to reach consensus on the provisional forward translation. Then, two other back translators were blind to the source language scales. Finally, a consensus was reach with the translation team.
The PCPI-S consists of 17 dimensions with 59 items about the three domains of the theoretical framework: prerequisites, care environment, and person-centred process. The prerequisites include five constructs: being professionally competent (Q1-Q3), developing interpersonal skills (Q4-Q7), showing commitment to work (Q8-Q12), knowing oneself (Q13-Q15), and being able to clearly demonstrate one’s beliefs and values (Q16-Q18) [ 27 ]. The care environment comprises seven constructs: appropriate skill mix (Q19-Q21), shared decision-making system (Q22-Q25), effective relationships between team members (Q26-Q28), power sharing (Q29-Q32), potential for innovation and risk-taking (Q33-Q35), physical environment (Q36-Q38), and supportive organisational system (Q39-Q43) [ 27 ]. The person-centred processes have five constructs: working with the patient’s beliefs and values (Q44-Q47), shared decision-making (Q48-Q50), authentic engagement in the relationship (Q51-Q53), being present with caring (Q54-Q56), and working holistically with the whole person (Q57-Q59).
Items are scored on a 5-point Likert scale ranging from 1 (“strongly disagree”) to 5 (“strongly agree”). The score for each construct is obtained by averaging the total items in the construct. The total score is obtained by averaging the scores of the constructs. Pearson’s correlation coefficient is used to calculate the correlations between the three main domains of the PCPI-S (prerequisites, care environment, and person-centred process).
The PCPI-C comprises 18 items aimed at evaluating patients’ agreement levels with statements regarding the person-centred process dimensions described in the PCPF. The PCPI-C comprises five constructs: working with the person’s beliefs and values (Q1-14-7-6), sharing decision-making (Q3-17-20-10), engaging authentically (Q12-18-9), being sympathetically present (Q16-5-2), and working holistically (Q 15-8-4-19). The PCPI-C uses a 5-point Likert scale ranging from 1 (“strongly disagree”) to 5 (“strongly agree”). The score for each construct is obtained by averaging the scores of the items in the construct. The total score is obtained by averaging the scores of the constructs.
The following characteristics were collected from health care staff: gender, age, profession, level of training, additional training, years of experience, care unit, activity rate, and years of experience in the current unit. Patients characteristics were retrieved from health electronic records and included gender, age, length of hospital stay at the time of completing the PCPI-C and whether patients were in single or shared room as it could influence the perception of care environment.
Participants
All health care staff members from participating units who were directly involved in patient care were invited to participate in completing the PCPI-S. A sample of patient participants was recruited on a voluntary basis from the participating units. Inclusion criteria for patients included being 18 years or older, proficient in reading and understanding French, and deemed cognitively capable by the healthcare team to complete the PCPI-C. The target sample size was 600 healthcare staff members and 200 patients to meet the criteria defined by the COSMIN [ 18 ].
Data collection
An email containing the URL to access the online PCPI-S was sent to healthcare staff members within the participating units. A data collection day was organized at each participating unit in the six hospitals. During this day, eligible patients were identified by the healthcare team. The study’s purpose and questionnaire were orally explained to the participants by the researcher. For participants capable of completing it independently, the PCPI-C paper questionnaire was provided and collected after completion at the end of the day. For patients who were unable to complete the questionnaire due to visual or motor impairments, the researcher either assisted in reading the questionnaire or provided physical support. The researcher paid careful attention to reading the questionnaire faithfully and avoiding influencing the participants’ responses.
Statistical analysis
Descriptive statistical analyses of the instruments and participants’ characteristics were performed by calculating mean and standard deviation.
For assessing psychometric properties, confirmatory factor analysis (CFA) was performed based on the structure of the PCPF theoretical framework. The parameters of the structural equation model were estimated by using the maximum likelihood ratio method. Missing data were left in the analyses and the maximum likelihood with missing (MLMV) model was used in Structural Equation Modelling. The internal consistency of the instruments was determined by using the McDonald’s Omega coefficient. The Omega coefficient can be judged as acceptable at over 0.70. Model fits were assessed using three fit indices and their goodness of fit criteria: root mean square error of approximation (RMSEA) (< 0.08), comparative fit index (CFI) (> 0.90), and standardized root mean square residuals (SRMR) (< 0.08). At least one of these criteria should be met to support the construct validity [ 28 ]; if the non-centrality index, RMSEA, is > 90%; and if its parsimony index, the Akaike information criterion, is lowest [ 29 ]. Analyses were performed by using Stata/IC software 17 [ 30 ].
Ethical considerations
The study was submitted and approved by the ethics committee of the canton of Vaud (CER-VD 2020 − 01562). All participants were informed about the study and gave consent to participate.
Participant characteristics
A total sample of 558 healthcare staff members completed the PCPI-S. They were predominantly women (85%) and worked as nurses (62%). Most staff members worked in medical (33%) and surgical wards (15%). Patient participants ( n = 510) were 70 years old on average and women accounted for half of the sample (51%). The mean length of stay when completing the PCPI-C was 11 days. Patients were mostly hospitalised in medical (36%) and surgical wards (27%) (Table 1 ).
- Psychometric analysis
All items of the PCPI-S and PCPI-C received positive scores, with mean scores ranging from 2.49 to 4.54. For the patient sample, there were 4 missing responses (0.8%) for questions 1 and 3, to 14 missing responses (2.8%) for questions 19 and 20. For the caregiver sample, there were 1 missing responses (0.2%) for questions 2 to 6, up to 68 missing responses (15%) for questions 44 to 59. Pearson’s correlation coefficient indicates statistically significant positive correlations between the three main domains of the PCPI-S: prerequisites and care environment ( r = 0.57, p < 0.01), prerequisites and person-centred process ( r = 0.72, p < 0.01), and care environment and person-centred process ( r = 0.49, p < 0.01). Factor loadings ranged from 0.35 to 0.89, with the majority exceeding 0.5. Notably, all factor loadings were statistically significant (standard error < 0.9; p < 0.01) and made meaningful contributions to the measurement model. As a result, these items were retained in the analysis [ 31 ]. Detailed factor loadings are presented in additional files 1 and 2.
In the case of the PCPI-S scale, the Omega coefficients for each domain were deemed acceptable, ranging from 0.87 for the Prerequisites factor to 0.93 for Person-centred processes. The specific Omega coefficients for each factor can be found in additional files 1 and 2. Regarding the PCPI-C scale, the Omega coefficients for each construct were also found to be acceptable, ranging from 0.64 for the Engaging Authenticity factor to 0.74 for Patient Beliefs and Values. The Omega coefficients for each factor are detailed in additional files 1 and 2. Furthermore, the Omega coefficient for the person-centred processes domain was outstanding, scoring at 0.92.
The model fit statistics of the three constructs indicated a good model fit, with a RMSEA close to 0.06, a 90% higher bracket below 0.09, a CFI of 0.90 or higher, and an SRMR less than 0.08. The detailed scores are set out in Table 2 .
The results of the psychometric analysis of the PCPI-S demonstrate good construct validity and internal consistency, thereby confirming the underlying principles of the theoretical PCP Framework. The model fit statistics consistently indicate a good fit for the three constructs within the PCPI-S. The PCPI-C demonstrates a reasonable to acceptable fit, indicating that while the model is not perfect, it is sufficiently robust for practical applications.
Examining the psychometric properties across different linguistic versions of the PCPI-S provides valuable insights into the instrument’s consistency and internal consistency across diverse cultural and linguistic contexts. In the present study, the Omega coefficient values for the PCPI-S and PCPI-C were consistently above 0.70, indicating robust internal consistency. The results for the PCPI-S are in line with previous research conducted in Swiss German, Austrian, Norwegian, Malaysian and Portuguese studies, which reported high Cronbach’s alpha scores (a > 0.70) [ 19 – 23 , 25 ]. These findings confirm the instrument’s strong internal consistency when measuring person-centred care constructs.
Regarding the RMSEA values, the PCPI-S versions in Swiss German, German, Norwegian, and Malaysian studies consistently indicated a good model fit, with RMSEA values ranging from 0.041 to 0.078 [ 19 – 22 ]. All these values were close to 0.06, indicating a good model fit. In the PCPI-S French version, RMSEA values ranged from 0.000 to 0.078. Although the RMSEA for prerequisites and the care environment was slightly higher in the French version than in the Swiss German and Norwegian versions, the RMSEA for person-centred processes was notably lower, suggesting an good fit for this construct in the present study.
The CFI values for the PCPI-S were generally above 0.90 across different linguistic versions, supporting the instrument’s construct validity and internal consistency. In the present study, the CFI values ranged from 0.85 for the care environment to 1.00 for person-centred processes, indicating an excellent fit for this construct. However, the slightly lower CFI for the care environment was consistent with findings in other studies.
The variations observed in different studies across languages may be attributed to linguistic nuances, cultural differences, or contextual factors specific to each linguistic group. These differences highlight the importance of adapting the instruments to the cultural and linguistic context in which they are used, emphasizing the ongoing need for validation and adaptation efforts. The findings from translations into French, Swiss German, German, Norwegian, and Malaysian languages collectively underscore the robustness and adaptability of the PCPI as a tool for assessing person-centred practice in diverse cultural contexts. The consistently high Cronbach’s alpha scores, meaningful factor loadings, and favourable GFIs in these translations suggest that the PCPI maintains its internal consistency and construct validity when applied in different linguistic and cultural settings.
Implications for clinical practice and future research
The PCPI has demonstrated strong internal consistency and good model fit across different linguistic versions. While the French-translated PCPI-S shows promising construct validity, its length may pose a challenge for widespread clinical adoption. Considering the time constraints frequently encountered in healthcare settings, there is a need for future research to design a shorter yet psychometrically robust version of the scale. This would enable quicker and more efficient assessments of patient-centred care without compromising measurement quality.
The availability of instruments aligned with a theoretical person-centred framework provides healthcare staff with a standardised measure to evaluate the degree of alignment with person-centered principles in care delivery. Consistent use of the PCPI-S and PCPI-C enable healthcare staff to collectively identify areas that require improvement, thereby fostering a continuous quality improvement process. Furthermore, insights gained from the PCPI-S and PCPI-C could inform the development of training programs aimed at enhancing person-centered care competencies among healthcare professionals.
Strengths and limitations
The large participation of both healthcare staff members and patient from six hospital and multiple clinical settings enhance generalisability of the results and confidence in the findings. Nonetheless, certain limitations should be acknowledged. The sample predominantly comprising nurses, and the relative homogeneity in participants’ responses could suggest a limited familiarity with the person-centred principles and the PCPF among both professionals and patients. While this study used CFA for psychometric analysis, further psychometric validation of the PCPI-S in French should include additional analyses such as a test-retest procedure and concurrent validity assessment. Finally, as professionals and patients participated on a voluntary basis, we cannot exclude a potential selection and desirability bias.
The psychometric analysis conducted in this study indicates high construct validity and internal consistency for the French translation of both the PCPI-S and the PCPI-C. The results presented in this article will enable international comparative studies and support the further development of person-centred care in French-speaking clinical settings.
Data availability
The datasets used and analysed during the current study are available on www.zenodo.org. DOI: 10.5281/zenodo.10849449.
Abbreviations
Person-centred Practice Inventory – Staff
Person-centred Practice Inventory – Care
- Confirmatory factor analysis
Person-centred Practice Framework
Consensus-based Standards for the selection of health Measurement Instruments
Root mean square error of approximation
Standardised root mean residual
Comparative fit index
Goodness-of-fit index
Confidence interval
McCormack B, McCance T, Bulley C, Brown D, McMillan A, Martin S. Fundamentals of person-centred healthcare practice. 1; Wiley Blackwell; 2021.
Kitson A, Marshall A, Bassett K, Zeitz K. What are the core elements of patient-centred care? A narrative review and synthesis of the literature from health policy, medicine and nursing. J Adv Nurs. 2013;69:4–15. https://doi.org/10.1016/j.ijnurstu.2017.11.007
McCormack B, Borg M, Cardiff S, Dewing J, Jacobs G, Janes N, et al. Person-centredness – the ‘state’ of the art. Int Pract Dev J. 2015;5.
Rathert C, Wyrwich MD, Boren SA. Patient-centered care and outcomes:a systematic review of the literature. Med Care Res Rev. 2013;70:351–79. https://doi.org/10.1177/1077558712465774 .
Olsson LE, Jakobsson Ung E, Swedberg K, Ekman I. Efficacy of person-centred care as an intervention in controlled trials – a systematic review. J Clin Nurs. 2013;22:456–65. https://doi.org/10.1111/jocn.12039 .
McMillan SS, Kendall E, Sav A, King MA, Whitty JA, Kelly F, et al. Patient-centered approaches to health care: a systematic review of randomized controlled trials. Med Care Res Rev. 2013;70:567–96. https://doi.org/10.1177/1077558713496318 .
Sidani S. Effects of patient-centered care on patient outcomes: an evaluation. Res Theory Nurs Pract. 2008;22:24–37.
Bachnick S, Ausserhofer D, Baernholdt M, Simon M. Patient-centered care, nurse work environment and implicit rationing of nursing care in Swiss acute care hospitals: a cross-sectional multi-center study. Int J Nurs Stud. 2018;81:98–106. https://doi.org/10.1016/j.ijnurstu.2017.11.007 .
Stone S. A retrospective evaluation of the impact of the planetree patient-centered model of care on inpatient quality outcomes. HERD: J. Environ. Health Res Des J. 2008;1:55–69. https://doi.org/10.1177/193758670800100406 .
Almalki ZS, Alotaibi AA, Alzaidi WS, Alghamdi AA, Bahowirth AM, Alsalamah NM. Economic benefits of implementing patient-centered medical home among patients with hypertension. Clinicoecon Outcomes Res. 2018;10:665–73. https://doi.org/10.2147/ceor.S179337 .
Park M, Giap TT, Lee M, Jeong H, Jeong M, Go Y. Patient- and family-centered care interventions for improving the quality of health care: a review of systematic reviews. Int J Nurs Stud. 2018;87:69–83. https://doi.org/10.1016/j.ijnurstu.2018.07.006 .
Janerka C, Leslie GD, Gill FJ. Development of patient-centred care in acute hospital settings: a metanarrative review. Int J Nurs Stud. 2023;140:104465. https://doi.org/10.1016/j.ijnurstu.2023.104465 .
McCormack B. Person-centred care and measurement: the more one sees, the better one knows where to look. J Health Serv Res Pol. 2022;27:85–7. https://doi.org/10.1177/13558196211071041 .
Edvardsson D, Innes A. Measuring person-centered care: a critical comparative review of published tools. Gerontologist. 2010;50:834–46. https://doi.org/10.1093/geront/gnq047 .
McCormack B. Person-centred care and measurement: the more one sees, the better one knows where to look. J Health Serv Res Policy. 2022;27:85–7. https://doi.org/10.1177/13558196211071041 .
Slater P, McCance T, McCormack B. The development and testing of the person-centred practice inventory – staff (PCPI-S). Int J Qual Health Care. 2017;29:541–7. https://doi.org/10.1093/intqhc/mzx066 .
(PCP-ICoP). TP-cPICoPC. Person-centred practice international community of practice version 1 2024. [Available from: http://www.pcp-icop.org ].
Mokkink LB, Terwee CB, Patrick DL, Alonso J, Stratford PW, Knol DL, et al. The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J clin epidemiol. 2010;63:737–45. https://doi.org/10.1016/j.jclinepi.2010.02.006 .
von Dach C, Schlup N, Gschwenter S, McCormack B. German translation, cultural adaptation and validation of the person-centred practice inventory—staff (PCPI-S). BMC Health Serv Res. 2023;23:458. https://doi.org/10.1186/s12913-023-09483-8 .
Bing-Jonsson PC, Slater P, McCormack B, Fagerström L. Norwegian translation, cultural adaption and testing of the Person-centred Practice Inventory – Staff (PCPI-S). BMC Health Services Research. 2018;18:555. https://doi.org/10.1186/s12913-018-3374-5 .
Weis MLD, Wallner M, Köck-Hódi S, Hildebrandt C, McCormack B, Mayer H. German translation, cultural adaptation and testing of the person-centred practice inventory - staff (PCPI-S). Nurs Open. 2020;7:1400–11. https://doi.org/10.1002/nop2.511 .
Balqis-Ali NZ, Saw PS, Jailani AS, Fun WH, Mohd Saleh N, Tengku Bahanuddin TPZ, et al. Cross-cultural adaptation and exploratory factor analysis of the person-centred practice inventory - staff (PCPI-S) questionnaire among Malaysian primary healthcare providers. BMC Health Serv Res. 2021;21:32. https://doi.org/10.1186/s12913-020-06012-9 .
Balqis-Ali NZ, Saw PS, Anis-Syakira J, Fun WH, Sararaks S, Lee SWH, et al. Healthcare provider personcentred practice: relationships between prerequisites, care environment and care processes using structural equation modelling. BMC Health Serv Res. 2022;22:576. https://doi.org/10.1186/s12913-022-07917-3 .
Errasti-Ibarrondo B, La Rosa-Salas V, Lizarbe-Chocarro M, Gavela-Ramos Y, Choperena A, Arbea Moreno L, et al. [Translation and transcultural adaptation of the Person-Centred Practice Inventory Staff (PCPI-S) for health professionals in Spain]. An Sist Sanit Navar. 2023;46. https://doi.org/10.23938/assn.1039 .
Ventura F, Costa P, Chaplin J, Domingues I, Jorge de Oliveira Ferreira R, McCormack B, et al. Portuguese translation, cultural adaptation, and validation of the Person-Centred Practice Inventory - Staff (PCPI-S). Ciência Saûde Coletiva. 2023;28. https://doi.org/10.1590/1413-812320232811.17072022 .
Wild D, Grove A, Martin M, Eremenco S, McElroy S, Verjee-Lorenz A, et al. Principles of good practice for the translation and cultural adaptation process for patient-reported outcomes (PRO) Measures: Report of the ISPOR Task Force for Translation and Cultural Adaptation. Value in Health. 2005;8:94–104. https://doi.org/10.1111/j.1524-4733.2005.04054.x .
Slater E, McCormack B, McCance T. The person centred practice inventory – staff score sheet.
Prinsen CAC, Mokkink LB, Bouter LM, Alonso J, Patrick DL, de Vet HCW, et al. COSMIN guideline for systematic reviews of patient-reported outcome measures. Qual Life Res. 2018;27:1147–57. https://doi.org/10.1007/s11136-018-1798-3 .
Hoe S. Issues and procedures in adopting structural equation modelling technique. J Qual Methods 2008;3:76–83.
StataCorp. Stata Statistical Software: Release 17. College Station, TX: StataCorporation; 2021.
Weiber R, Sarstedt M. Strukturgleichungsmodellierung. Eine anwendungsorientierte Einführung in die Kausalanalyse mit Hilfe von AMOS, SmartPLS und SPSS: Springer Gabler; 2021.
Download references
Acknowledgements
The authors would like to thank all the healthcare professionals and patients who gave their time to participate in this study.
Not applicable.
Open access funding provided by University of Lausanne
Author information
Authors and affiliations.
Institute of Higher Education and Research in Healthcare (IUFRS), Lausanne University Hospital, University of Lausanne, Lausanne, Switzerland
Cedric Mabire & Joanie Pellet
Geneva Institution for Home Care and Assistance (IMAD), Geneva, Switzerland
Marie Piccot-Crezollet
Sydney Nursing School, Faculty of Medicine and Health, University of Sydney, Camperdown, NSW, Australia
Vaibhav Tyagi & Brendan McCormack
You can also search for this author in PubMed Google Scholar
Contributions
Data collection: MP-C collected data from participants and coordinated the study on hospital sites. Data analysis and interpretation: CM conducted the psychometric analysis. Drafting the article: CM, MP-C, and JP drafted the manuscript. Critical revision of the manuscript: VT and BM. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Cedric Mabire .
Ethics declarations
Ethics approval and consent to participate, consent for publication, competing interests.
The authors declare no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Mabire, C., Piccot-Crezollet, M., Tyagi, V. et al. Structural validation of two person-centred practice inventories PCPI-S and PCPI-C - French version. BMC Health Serv Res 24 , 1092 (2024). https://doi.org/10.1186/s12913-024-11432-y
Download citation
Received : 08 December 2023
Accepted : 13 August 2024
Published : 18 September 2024
DOI : https://doi.org/10.1186/s12913-024-11432-y
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Person-centred care
BMC Health Services Research
ISSN: 1472-6963
- General enquiries: [email protected]
Advertisement
Chinese utility weights for the EORTC cancer-specific utility instrument QLU-C10D
- Published: 13 September 2024
Cite this article

- Yiyin Cao 1 na1 ,
- Juan Xu 1 , 2 na1 ,
- Richard Norman 3 ,
- Madeleine T. King 4 ,
- Georg Kemmler 5 ,
- Weidong Huang ORCID: orcid.org/0000-0001-8156-5537 1 &
- Nan Luo 6
The aim of this study is to provide Chinese utility weights for the European Organization for Research and Treatment of Cancer Quality of Life Utility Measure-Core 10 Dimensions (EORTC QLU-C10D) which is a preference-based cancer-specific utility instrument derived from the EORTC QLQ-C30.
We conducted an online survey of the general population in China, with quota sampling for age and gender. Each respondent was asked to complete a discrete choice experimental survey consisting of 16 randomly selected choice sets. The conditional logit model and mixed logit model were used to analyze respondents’ preferences, and the goodness of fit of the model was tested.
A total of 2003 respondents were included in the analysis. Utility decrements within dimensions were typically monotonic. Monotonic inconsistency issues in the Fatigue, Sleep, and Nausea dimensions were normalized by monotonicity correction. Physical functioning, Pain, and Role functioning were associated with the greatest utility weights, with the smallest decrements being in Bowel problems and Emotional functioning. The utility value for the worst health state was 0.083, i.e. slightly higher than being dead.
Conclusions
This study provides the first China-specific set of value for the QLU-C10D based on societal preferences of the Chinese adult general population. The value set can be used as a cancer-specific scoring system for economic evaluations of new oncology therapies and technologies in China.
Plain summary
This study provides the first China-specific set of value for the QLU-C10D based on societal preferences of the Chinese adult general population.
The value set can be used as a cancer-specific scoring system for economic evaluations of new oncology therapies and technologies in China.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Subscribe and save.
- Get 10 units per month
- Download Article/Chapter or eBook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
Price includes VAT (Russian Federation)
Instant access to the full article PDF.
Rent this article via DeepDyve
Institutional subscriptions

Similar content being viewed by others

Dutch utility weights for the EORTC cancer-specific utility instrument: the Dutch EORTC QLU-C10D
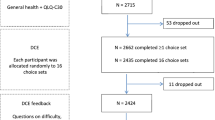
Japanese value set for the EORTC QLU-C10D: A multi-attribute utility instrument based on the EORTC QLQ-C30 cancer-specific quality-of-life questionnaire
Australian utility weights for the eortc qlu-c10d, a multi-attribute utility instrument derived from the cancer-specific quality of life questionnaire, eortc qlq-c30, explore related subjects.
- Medical Ethics
Siegel, R. L., Miller, K. D., Fuchs, H. E., & Jemal, A. (2021). Cancer statistics, 2021. C Ca: A Cancer Journal for Clinicians , 71 (1), 7–33. https://doi.org/10.3322/caac.21654
Article Google Scholar
Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., & Bray, F. (2021). Global Cancer statistics 2020: GLOBOCAN estimates of incidence and Mortality Worldwide for 36 cancers in 185 countries. C Ca: A Cancer Journal for Clinicians , 71 (3), 209–249. https://doi.org/10.3322/caac.21660
Article CAS Google Scholar
McGuire, S., & WHO Press. (2016). World Cancer Report 2014. Geneva, Switzerland: World Health Organization, International Agency for Research on Cancer, 2015. Adv Nutr , 7 (2), 418–419. https://doi.org/10.3945/an.116.012211
World Health Organization. (2020). World cancer report: Cancer research for cancer prevention . International Agency for Research on Cancer.
Zheng, R. S., Zhang, S. W., Sun, K. X., Chen, R., Wang, S. M., Li, L., Zeng, H. M., Wei, W. W., & He, J. (2023). [Cancer statistics in China, 2016]. Zhonghua Zhong Liu Za Zhi , 45 (3), 212–220. https://doi.org/10.3760/cma.j.cn112152-20220922-00647
Article CAS PubMed Google Scholar
United Nations. World population prospects. New York: United Nations (2022). https://population.un.org/wpp/
Ferlay, J., Soerjomataram, I., Dikshit, R., Eser, S., Mathers, C., Rebelo, M., Parkin, D. M., Forman, D., & Bray, F. (2015). Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. International Journal of Cancer , 136 (5), E359–386. https://doi.org/10.1002/ijc.29210
DiMasi, J. A., Grabowski, H. G., & Hansen, R. W. (2016). Innovation in the pharmaceutical industry: New estimates of R&D costs. Journal of Health Economics , 47 , 20–33. https://doi.org/10.1016/j.jhealeco.2016.01.012
Article PubMed Google Scholar
Elkin, E. B., Weinstein, M. C., Winer, E. P., Kuntz, K. M., Schnitt, S. J., & Weeks, J. C. (2004). HER-2 testing and trastuzumab therapy for metastatic breast cancer: A cost-effectiveness analysis. Journal of Clinical Oncology , 22 (5), 854–863. https://doi.org/10.1200/jco.2004.04.158
Khalili, F., Najafi, B., Mansour-Ghanaei, F., Yousefi, M., Abdollahzad, H., & Motlagh, A. (2020). Cost-effectiveness analysis of Colorectal Cancer Screening: A systematic review. Risk Manag Healthc Policy , 13 , 1499–1512. https://doi.org/10.2147/rmhp.S262171
Article PubMed PubMed Central Google Scholar
Clark, T. J., Barton, P. M., Coomarasamy, A., Gupta, J. K., & Khan, K. S. (2006). Investigating postmenopausal bleeding for endometrial cancer: Cost-effectiveness of initial diagnostic strategies. Bjog , 113 (5), 502–510. https://doi.org/10.1111/j.1471-0528.2006.00914.x
Hu, S., Gu, S., Qi, C., Wang, S., Qian, F., Shi, C., & Fan, G. (2023). Cost-utility analysis of semaglutide for type 2 diabetes after its addition to the National Medical Insurance System in China. Diabetes, Obesity & Metabolism , 25 (2), 387–397. https://doi.org/10.1111/dom.14881
World Health Organization. WHO Guideline on Country Pharmaceutical Pricing Policies. Geneva: World Health Organization (2020). https://www.who.int/publications/i/item/9789240011878
Yu, H., Zhang, H., Yang, J., Liu, C., Lu, C., Yang, H., Huang, W., Zhou, J., Fu, W., Shi, L., Yan, Y., Liu, G., & Li, L. (2018). Health utility scores of family caregivers for leukemia patients measured by EQ-5D-3L: A cross-sectional survey in China. Bmc Cancer , 18 (1), 950. https://doi.org/10.1186/s12885-018-4855-y
National Institute for Health and Care Excellence. Guide to the Methods of Technology Appraisal 2013. London: National Institute for Health and Care Excellence (NICE) (2013). https://www.nice.org.uk/process/pmg9
Canadian Agency for Drugs and Technologies in Health Guidelines for the Economic Evaluation of Health Technologies: Canada. Ottawa: Canadian Agency for Drugs and Technologies in Health (CADTH). https://www.cadth.ca/
Haute Autorité de Santé Choices in Methods for Economic Evaluation - A Methodological Guide. Saint-Denis La Plaine: Haute Autorité de Santé (HAS). https://www.has-sante.fr/
Drummond, M. F., Aguiar-Ibanez, R., & Nixon, J. (2006). Economic evaluation. Singapore Medical Journal , 47 (6), 456–461. quiz 462.
CAS PubMed Google Scholar
Garau, M., Shah, K. K., Mason, A. R., Wang, Q., Towse, A., & Drummond, M. F. (2011). Using QALYs in cancer: A review of the methodological limitations. Pharmacoeconomics , 29 (8), 673–685. https://doi.org/10.2165/11588250-000000000-00000
King, M. T., Norman, R., Mercieca-Bebber, R., Costa, D. S. J., McTaggart-Cowan, H., Peacock, S., Janda, M., Müller, F., Viney, R., Pickard, A. S., & Cella, D. (2021). The Functional Assessment of Cancer Therapy eight dimension (FACT-8D), a Multi-attribute Utility Instrument Derived from the Cancer-Specific FACT-General (FACT-G) quality of Life Questionnaire: Development and Australian Value Set. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 24 (6), 862–873. https://doi.org/10.1016/j.jval.2021.01.007
Gibson, A. E. J., Longworth, L., Bennett, B., Pickard, A. S., & Shaw, J. W. (2024). Assessing the content validity of preference-based measures in Cancer. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 27 (1), 70–78. https://doi.org/10.1016/j.jval.2023.10.006
López-Bastida, J., Oliva, J., Antoñanzas, F., García-Altés, A., Gisbert, R., Mar, J., & Puig-Junoy, J. (2010). Spanish recommendations on economic evaluation of health technologies. The European Journal of Health Economics , 11 (5), 513–520. https://doi.org/10.1007/s10198-010-0244-4
King, M. T., Costa, D. S., Aaronson, N. K., Brazier, J. E., Cella, D. F., Fayers, P. M., Grimison, P., Janda, M., Kemmler, G., Norman, R., Pickard, A. S., Rowen, D., Velikova, G., Young, T. A., & Viney, R. (2016). QLU-C10D: A health state classification system for a multi-attribute utility measure based on the EORTC QLQ-C30. Quality of Life Research , 25 (3), 625–636. https://doi.org/10.1007/s11136-015-1217-y
King, M. T., Viney, R., Simon Pickard, A., Rowen, D., Aaronson, N. K., Brazier, J. E., Cella, D., Costa, D. S. J., Fayers, P. M., Kemmler, G., McTaggart-Cowen, H., Mercieca-Bebber, R., Peacock, S., Street, D. J., Young, T. A., & Norman, R. (2018). Australian utility weights for the EORTC QLU-C10D, a Multi-attribute Utility Instrument Derived from the Cancer-Specific Quality of Life Questionnaire, EORTC QLQ-C30. Pharmacoeconomics , 36 (2), 225–238. https://doi.org/10.1007/s40273-017-0582-5
McTaggart-Cowan, H., King, M. T., Norman, R., Costa, D. S. J., Pickard, A. S., Regier, D. A., Viney, R., & Peacock, S. J. (2019). The EORTC QLU-C10D: The Canadian valuation study and algorithm to Derive Cancer-Specific Utilities from the EORTC QLQ-C30. MDM Policy Pract , 4 (1), 2381468319842532. https://doi.org/10.1177/2381468319842532
Kemmler, G., Gamper, E., Nerich, V., Norman, R., Viney, R., Holzner, B., & King, M. (2019). German value sets for the EORTC QLU-C10D, a cancer-specific utility instrument based on the EORTC QLQ-C30. Quality of Life Research , 28 (12), 3197–3211. https://doi.org/10.1007/s11136-019-02283-w
Norman, R., Mercieca-Bebber, R., Rowen, D., Brazier, J. E., Cella, D., Pickard, A. S., Street, D. J., Viney, R., Revicki, D., & King, M. T. (2019). U.K. utility weights for the EORTC QLU-C10D. Health Economics , 28 (12), 1385–1401. https://doi.org/10.1002/hec.3950
Gamper, E. M., King, M. T., Norman, R., Efficace, F., Cottone, F., Holzner, B., & Kemmler, G. (2020). EORTC QLU-C10D value sets for Austria, Italy, and Poland. Quality of Life Research , 29 (9), 2485–2495. https://doi.org/10.1007/s11136-020-02536-z
Article CAS PubMed PubMed Central Google Scholar
Nerich, V., Gamper, E. M., Norman, R., King, M., Holzner, B., Viney, R., & Kemmler, G. (2021). French Value-Set of the QLU-C10D, a Cancer-specific utility measure derived from the QLQ-C30. Applied Health Economics and Health Policy , 19 (2), 191–202. https://doi.org/10.1007/s40258-020-00598-1
Jansen, F., Verdonck-de Leeuw, I. M., Gamper, E., Norman, R., Holzner, B., King, M., & Kemmler, G. (2021). Dutch utility weights for the EORTC cancer-specific utility instrument: The Dutch EORTC QLU-C10D. Quality of Life Research , 30 (7), 2009–2019. https://doi.org/10.1007/s11136-021-02767-8
Revicki, D. A., King, M. T., Viney, R., Pickard, A. S., Mercieca-Bebber, R., Shaw, J. W., Müller, F., & Norman, R. (2021). United States Utility Algorithm for the EORTC QLU-C10D, a Multiattribute Utility Instrument based on a Cancer-specific quality-of-life instrument. Medical Decision Making , 41 (4), 485–501. https://doi.org/10.1177/0272989x211003569
Finch, A. P., Gamper, E., Norman, R., Viney, R., Holzner, B., King, M., & Kemmler, G. (2021). Estimation of an EORTC QLU-C10 value set for Spain using a Discrete Choice Experiment. Pharmacoeconomics , 39 (9), 1085–1098. https://doi.org/10.1007/s40273-021-01058-x
Lehmann, J., Rojas-Concha, L., Petersen, M. A., Holzner, B., Norman, R., King, M. T., & Kemmler, G. (2024). Danish value sets for the EORTC QLU-C10D utility instrument. Quality of Life Research , 33 (3), 831–841. https://doi.org/10.1007/s11136-023-03569-w
Shiroiwa, T., King, M. T., Norman, R., Müller, F., Campbell, R., Kemmler, G., Murata, T., Shimozuma, K., & Fukuda, T. (2024). Japanese value set for the EORTC QLU-C10D: A multi-attribute utility instrument based on the EORTC QLQ-C30 cancer-specific quality-of-life questionnaire. Quality of Life Research , 33 (7), 1865–1879. https://doi.org/10.1007/s11136-024-03655-7
Luo, N., Liu, G., Li, M., Guan, H., Jin, X., & Rand-Hendriksen, K. (2017). Estimating an EQ-5D-5L value set for China. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 20 (4), 662–669. https://doi.org/10.1016/j.jval.2016.11.016
Liu, G. G., Wu, H., Li, M., Gao, C., & Luo, N. (2014). Chinese time trade-off values for EQ-5D health states. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 17 (5), 597–604. https://doi.org/10.1016/j.jval.2014.05.007
Zhuo, L., Xu, L., Ye, J., Sun, S., Zhang, Y., Burstrom, K., & Chen, J. (2018). Time Trade-Off Value set for EQ-5D-3L based on a nationally Representative Chinese Population Survey. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 21 (11), 1330–1337. https://doi.org/10.1016/j.jval.2018.04.1370
Liu, G. G., Guan, H., Jin, X., Zhang, H., Vortherms, S. A., & Wu, H. (2022). Rural population’s preferences matter: A value set for the EQ-5D-3L health states for China’s rural population. Health and Quality of Life Outcomes , 20 (1), 14. https://doi.org/10.1186/s12955-022-01917-x
Yang, Z., Jiang, J., Wang, P., Jin, X., Wu, J., Fang, Y., Feng, D., Xi, X., Li, S., Jing, M., Zheng, B., Huang, W., & Luo, N. (2022). Estimating an EQ-5D-Y-3L value set for China. Pharmacoeconomics , 40 (Suppl 2), 147–155. https://doi.org/10.1007/s40273-022-01216-9
Wu, J., Xie, S., He, X., Chen, G., Bai, G., Feng, D., Hu, M., Jiang, J., Wang, X., Wu, H., Wu, Q., & Brazier, J. E. (2021). Valuation of SF-6Dv2 Health states in China using Time Trade-off and discrete-choice experiment with a duration dimension. Pharmacoeconomics , 39 (5), 521–535. https://doi.org/10.1007/s40273-020-00997-1
Peeters, Y., & Stiggelbout, A. M. (2010). Health state valuations of patients and the general public analytically compared: A meta-analytical comparison of patient and population health state utilities. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 13 (2), 306–309. https://doi.org/10.1111/j.1524-4733.2009.00610.x
Stiggelbout, A. M., & de Haes, J. C. (2001). Patient preference for cancer therapy: An overview of measurement approaches. Journal of Clinical Oncology , 19 (1), 220–230. https://doi.org/10.1200/jco.2001.19.1.220
Gamper, E. M., King, M. T., Norman, R., Loth, F. L. C., Holzner, B., & Kemmler, G. (2022). The EORTC QLU-C10D discrete choice experiment for cancer patients: A first step towards patient utility weights. J Patient Rep Outcomes , 6 (1), 42. https://doi.org/10.1186/s41687-022-00430-5
Gamper, E. M., Holzner, B., King, M. T., Norman, R., Viney, R., Nerich, V., & Kemmler, G. (2018). Test-retest reliability of Discrete Choice experiment for valuations of QLU-C10D Health states. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 21 (8), 958–966. https://doi.org/10.1016/j.jval.2017.11.012
Norman, R., Viney, R., Aaronson, N. K., Brazier, J. E., Cella, D., Costa, D. S., Fayers, P. M., Kemmler, G., Peacock, S., Pickard, A. S., Rowen, D., Street, D. J., Velikova, G., Young, T. A., & King, M. T. (2016). Using a discrete choice experiment to value the QLU-C10D: Feasibility and sensitivity to presentation format. Quality of Life Research , 25 (3), 637–649. https://doi.org/10.1007/s11136-015-1115-3
Mulhern, B., Norman, R., Street, D. J., & Viney, R. (2019). One method, many methodological choices: A structured review of Discrete-Choice experiments for Health State Valuation. Pharmacoeconomics , 37 (1), 29–43. https://doi.org/10.1007/s40273-018-0714-6
National Bureau of Statistics of China. (2021). China Statistical Yearbook . China Statistic Publishing House.
National Bureau of Statistics of China. (2020). The 2019 Population Census of the people’s Republic of China . China Statistic Publishing House.
United Nations Statistics Division. (2010). Population Censuses’ Datasets . United Nations.
Norman, R., Kemmler, G., Viney, R., Pickard, A. S., Gamper, E., Holzner, B., Nerich, V., & King, M. (2016). Order of Presentation of Dimensions Does Not Systematically Bias Utility Weights from a Discrete Choice Experiment. Value in health: the journal of the International Society for Pharmacoeconomics and Outcomes Research, 19(8), 1033–1038. http://www.99885.net/doi.php?doi=10.1016/j.jval.2016.07.003
National Health Commission of China. (2020). China Health Statistics Yearbook . Peking Union Medical College Publishing House.
Aaronson, N. K., Ahmedzai, S., Bergman, B., Bullinger, M., Cull, A., Duez, N. J., Filiberti, A., Flechtner, H., Fleishman, S. B., de Haes, J. C., et al. (1993). The European Organization for Research and Treatment of Cancer QLQ-C30: A quality-of-life instrument for use in international clinical trials in oncology. Journal of the National Cancer Institute , 85 (5), 365–376. https://doi.org/10.1093/jnci/85.5.365
Giesinger, J. M., Efficace, F., Aaronson, N., Calvert, M., Kyte, D., Cottone, F., Cella, D., & Gamper, E. M. (2021). Past and current practice of patient-reported outcome measurement in Randomized Cancer clinical trials: A systematic review. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 24 (4), 585–591. https://doi.org/10.1016/j.jval.2020.11.004
Gao, S., Corrigan, P. W., Qin, S., & Nieweglowski, K. (2019). Comparing Chinese and European American mental health decision making. Journal of Mental Health (Abingdon, England) , 28 (2), 141–147. https://doi.org/10.1080/09638237.2017.1417543
Scott, N. W., Fayers, P. M., Bottomley, A., Aaronson, N. K., de Graeff, A., Groenvold, M., Koller, M., Petersen, M. A., & Sprangers, M. A. (2006). Comparing translations of the EORTC QLQ-C30 using differential item functioning analyses. Qual Life Res , 15 (6), 1103–1115; discussion 1117–1120. https://doi.org/10.1007/s11136-006-0040-x
Shiroiwa, T., Ikeda, S., Noto, S., Igarashi, A., Fukuda, T., Saito, S., & Shimozuma, K. (2016). Comparison of Value Set based on DCE and/or TTO Data: Scoring for EQ-5D-5L Health states in Japan. Value In Health : The Journal of the International Society for Pharmacoeconomics and Outcomes Research , 19 (5), 648–654. https://doi.org/10.1016/j.jval.2016.03.1834
Yang, Z., van Busschbach, J., Timman, R., Janssen, M. F., & Luo, N. (2017). Logical inconsistencies in time trade-off valuation of EQ-5D-5L health states: Whose fault is it? PLoS One , 12 (9), e0184883. https://doi.org/10.1371/journal.pone.0184883
Jin, X., Liu, G. G., Luo, N., Li, H., Guan, H., & Xie, F. (2016). Is bad living better than good death? Impact of demographic and cultural factors on health state preference. Quality of Life Research , 25 (4), 979–986. https://doi.org/10.1007/s11136-015-1129-x
Download references
This work was supported by the National Natural Science Foundation of China (Grant No. 71974048, 72274045) and European Organization for Research and Treatment of Cancer under Grant (No. 002/2014).
Author information
Yiyin Cao and Juan Xu have made equal contributions and shared first authorship.
Authors and Affiliations
School of Health Management, Harbin Medical University, Harbin, China
Yiyin Cao, Juan Xu & Weidong Huang
Shenzhen Center, Cancer Hospital Chinese Academy of Medical Sciences, Shenzhen, China
School of Public Health, Curtin University, Perth, Australia
Richard Norman
Faculty of Science, School of Psychology, University of Sydney, Sydney, Australia
Madeleine T. King
Department of Psychiatry 1, Innsbruck Medical University, Innsbruck, Austria
Georg Kemmler
Saw Swee Hock School of Public Health, National University of Singapore, Singapore, Singapore
You can also search for this author in PubMed Google Scholar
Contributions
Concept and design: Weidong Huang, Nan Luo, Yiyin Cao and Juan Xu. Acquisition of data: Georg Kemmler, Weidong Huang. Analysis and interpretation of data: Richard Norman, Madeleine T. King and Georg Kemmler. Drafting of the manuscript: Yiyin Cao, Juan Xu, Weidong Huang, Richard Norman, Madeleine T. King and Georg Kemmler. Critical revision of paper for important intellectual content: Nan Luo, Richard Norman, Madeleine T. King and Georg Kemmler. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Correspondence to Weidong Huang .
Ethics declarations
Conflict of interest.
Madeleine King is the founding chair of the MAUCa Consortium. Richard Norman, Georg Kemmler and Nan Luo are members of MAUCa Consortium. Georg Kemmler and Madeleine T. King are members of EORTC QOL Group. As instrument developers, we are prone to bias towards our MAUI. All authors have no conflict of interest with each other.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the Ethics Committee of Harbin Medical University (project identification code: HMUIRB2023005).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Rights and permissions.
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Cao, Y., Xu, J., Norman, R. et al. Chinese utility weights for the EORTC cancer-specific utility instrument QLU-C10D. Qual Life Res (2024). https://doi.org/10.1007/s11136-024-03776-z
Download citation
Accepted : 24 August 2024
Published : 13 September 2024
DOI : https://doi.org/10.1007/s11136-024-03776-z
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Discrete choice experiment
- Health state valuation
- Find a journal
- Publish with us
- Track your research

IMAGES
VIDEO
COMMENTS
Survey Instruments in Research Methods. The following are some commonly used survey instruments in research methods: Questionnaires: A questionnaire is a set of standardized questions designed to collect information about a specific topic. Questionnaires can be administered in different ways, including in person, over the phone, or online.
Survey research means collecting information about a group of people by asking them questions and analyzing the results. To conduct an effective survey, follow these six steps: ... A questionnaire is a data collection tool or instrument, while a survey is an overarching research method that involves collecting and analyzing data from people ...
Survey Research. Definition: Survey Research is a quantitative research method that involves collecting standardized data from a sample of individuals or groups through the use of structured questionnaires or interviews. The data collected is then analyzed statistically to identify patterns and relationships between variables, and to draw conclusions about the population being studied.
Questionnaires vs. surveys. A survey is a research method where you collect and analyze data from a group of people. A questionnaire is a specific tool or instrument for collecting the data.. Designing a questionnaire means creating valid and reliable questions that address your research objectives, placing them in a useful order, and selecting an appropriate method for administration.
Survey research is defined as "the collection of information from a sample of individuals through their responses to questions" (Check & Schutt, 2012, p. 160). This type of research allows for a variety of methods to recruit participants, collect data, and utilize various methods of instrumentation. Survey research can use quantitative research ...
A research instrument is a tool you will use to help you collect, measure and analyze the data you use as part of your research. ... Surveys (online or in-person). In survey research, you are posing questions in which you ask for a response from the person taking the survey. You may wish to have either free-answer questions such as essay style ...
Survey Research Definition. Survey Research is defined as the process of conducting research using surveys that researchers send to survey respondents. The data collected from surveys is then statistically analyzed to draw meaningful research conclusions. In the 21st century, every organization's eager to understand what their customers think ...
Survey research is the process of collecting data from a predefined group (e.g. customers or potential customers) with the ultimate goal of uncovering insights about your products, services, or brand overall.. As a quantitative data collection method, survey research can provide you with a goldmine of information that can inform crucial business and product decisions.
Research instruments are measurement tools, such as questionnaires, scales, and surveys, that researchers use to measure variables in research studies. In most cases, it is better to use a previously validated instrument rather than create one from scratch. Always evaluate instruments for relevancy, validity, and reliability.
This is where a validated survey instrument comes in to the questionnaire design. Validated instruments are those that have been extensively tested and are correctly calibrated to their target. ... Survey research is a unique way of gathering information from a large cohort. Advantages of surveys include having a large population and therefore ...
Types of Research Instruments: Surveys Survey research encompasses any measurement procedures that involve asking questions of respondents. The types of surveys can vary on the span of time used to conduct the study. They can be comprised of cross-sectional surveys and/or longitudinal surveys. Types of questions asked in surveys include:
Research Instruments: Surveys, Questionnaires, and other Measurement Tools. This table is based on the work of Joanne Rich and Janet Schnall at the University of Washington Health Sciences Library. See their website for much more information on finding research instruments.
survey research. Pinsonneault and Kraemer (1993) defined a survey as a "means for gathering information about the characteristics, actions, or opinions of a large group of people" (p. 77). Surveys can also be used to assess needs, evaluate demand, and examine impact (Salant & Dillman, 1994, p. 2). The term . survey instrument. is often used ...
Definition: A Questionnaire is a research tool or survey instrument that consists of a set of questions or prompts designed to gather information from individuals or groups of people. It is a standardized way of collecting data from a large number of people by asking them a series of questions related to a specific topic or research objective.
What are Research Instruments? A research instrument is a tool used to collect, measure, and analyze data related to your subject. Research instruments can be tests, surveys, scales, questionnaires, or even checklists. To assure the strength of your study, it is important to use previously validated instruments! Getting Started.
Questionnaires can be used as the sole research instrument (such as in a cross sectional survey) or within clinical trials or epidemiological studies. Randomised trials are subject to strict reporting criteria, 4 but there is no comparable framework for questionnaire research.
Questionnaires are research instruments (surveys) consisting of a series of questions for the purpose of collecting information about a particular subject. 5 ... be sure to develop survey instruments for each, as respondents from each group might not be able to answer questions written for other groups. For example, youth
Sample size. Statistical power. Survey. Throughout our experience in research and supervising research students, two questions we are always consulted on after getting the research framework done are (1) how to collect the data, and (2) how many individuals do we need. Short answers for these questions are (1) design a survey, and (2) more is ...
In survey research, the instruments that are utilized can be either a questionnaire or an interview (either structured or unstructured). 1. Questionnaires. Typically, a questionnaire is a paper-and-pencil instrument that is administered to the respondents. The usual questions found in questionnaires are closed-ended questions, which are ...
Survey research is a method in which data is collected from a target population, called the sample, by personal interviews, online surveys, the telephone, ... Key Terms and Concepts: Survey instrument: The questionnaire or response item posed to a respondent is called a survey research instrument. The instrument may be a questionnaire or an ...
A questionnaire is a research instrument consisting of a series of questions for the purpose of gathering information from respondents. Questionnaires can be thought of as a kind of written interview. They can be carried out face to face, by telephone, computer, or post. Questionnaires provide a relatively cheap, quick, and efficient way of ...
A research instrument is a survey, questionnaire, test, scale, rating, or tool designed to measure the variable(s), characteristic(s), or information of interest, often a behavioral or psychological characteristic. Research instruments can be helpful tools to your research study.
The use of research questionnaires or surveys in nursing is a long standing tradition, dating back to the 1960s (Logan, 1966) and 1970s (Oberst, 1978), when the scientific discipline emerged.This type of tool enables nursing researchers to gather primary data from a specific population, whether it is patients, carers, nurses, or other stakeholders to address gaps in the existing evidence base ...
The model was fine-tuned on 3,000 labeled, open-ended responses from the NCHS Research and Development Survey's RANDS during COVID 19 Rounds 1 and 2 surveys. The base SimCSE BERT model was trained on BookCorpus and English Wikipedia. Training procedure. Learning rate: 5e-5. Batch size: 16. Number training epochs: 4. Base Model pooling dimension ...
Background The shift towards person-centred care has become integral in achieving high-quality healthcare, focusing on individual patient needs, preferences, and values. However, existing instruments for measuring person-centred practice often lack theoretical underpinnings and comprehensive assessment. The Person-centred Practice Inventory - Staff (PCPI-S) and the Person-centred Practice ...
Objective The aim of this study is to provide Chinese utility weights for the European Organization for Research and Treatment of Cancer Quality of Life Utility Measure-Core 10 Dimensions (EORTC QLU-C10D) which is a preference-based cancer-specific utility instrument derived from the EORTC QLQ-C30. Methods We conducted an online survey of the general population in China, with quota sampling ...