- Clinical Jobs
- Pharma Jobs
- Fresher jobs
- Walk-in Interviews
- Pharma Companies


Contract research organizations in Gurgaon
- Contract Research Organizations
- Current Page
| Address | 1st floor, Tower B & C, Building No. 8, DLF Cyber City Phase II |
| City name | Gurgaon – 122002 |
| Phone No | +91 124 452 0000 |
| Website | |
| Address | Ground to 7th floor, Building B1, DLF SEZ Zone, Sec 30, Vill :- Silokhera |
| City name | Gurgaon-122001 |
| Phone No | +91 124 4453 000 |
| Website | |
| Address | No. 404, BPTP Park Centra, NH -8, Sector – 30 |
| City name | Gurgaon – 122001 |
| Phone No | 91-124-4569000 |
| Website | |
| Address | 876, Udyog Vihar, Phase V, Gurgaon 122016, Haryana |
| City name | Gurgaon 122016 |
| Phone No | 91-8588857484 |
| Website | |
| Address | 2nd Floor, Tower-C, Building No.8,DLF Cyber City, DLF Phase-II, |
| City name | Gurgaon-122 002 |
| Phone No | 91-124-4500200 |
| Website | |
| Address | 12th Floor, Vatika Professional Point, Golf Course Extn Road, Badshahpur, Sector 66, |
| City name | Gurgaon-122001 |
| Phone No | 91 124 457 2300 |
| Website | |
| Address | 4th Floor, Plot no. 47, Sector – 44 |
| City name | Gurgaon – 122 002 |
| Phone No | 0124-4781400 |
| Website | |
| Address | Building No. 3, Unitech Realty Projects Limited, Ground, 1st & 2nd floor, Sector – 48, |
| City name | Gurgaon – 12200 |
| Phone No | 91 124 4413510 |
| Website | |
| Address | 7th Floor, Tower C, DLF Cyber Greens, Sector-25A, DLF City, Phase III |
| City name | Gurgaon – 122002 |
| Phone No | 91-124-4091818 |
| Website | |
| Address | Plot# 92, Sector 32, Institutional Area, |
| City name | Gurgaon-122001 |
| Phone No | 91-124-475300 |
| Website | |
| Address | Echelon Institutional Area, Plot No-11, Sector – 32 |
| City name | Gurgaon – 122001 |
| Phone No | 91 124 4885000 |
| Website | |
| Address | 1st Floor, Technopolis, Golf Course Road, Sector – 54, |
| City name | Gurgaon 122 001 |
| Phone No | 91(124) 4528300 |
| Website | |
| Address | 701 – 703 , Tower – B, ApexOne, Millennium Plaza, Near to HUDA city metro station. Sushant Lok – 1 |
| City name | Gurgaon – 122002 |
| Phone No | 91 124 3094000 |
| Website | |
| Address | DLF Building No. 10 C, 12th floor, Cyber City |
| City name | Gurgaon – 122 002 |
| Phone No | 0124 – 333 0100 |
| Website | |
| Address | Nestlé House, Jacaranda Marg M Block,DLF City Phase II, National Highway 8 |
| City name | Gurgaon 122 002 |
| Phone No | 91 124 238 93 00 |
| Website | |
| Address | 5th & 6th Floor, Technopolis, Tower B, Golf Course Road, Sector 54 |
| City name | Gurgaon 12202 |
| Phone No | 91 124 462 6356 |
| Website | |
| Address | Pfizer Centre, DLF Building 9B, 4th Floor, Tower B, DLF Cybercity, Phase III, |
| City name | Gurgaon – 122 002 |
| Phone No | 0124 – 302 3900 |
| Website | |
| Address | Village – Sehroul, Sector – 18, Udyog Vihar Industrial Area |
| City name | Gurgaon – 122015 |
| Phone No | 91 124 284 8500 |
| Website | |
| Address | Vatika City Point, 11th Floor, Sector 25, Mehrauli Gurgaon Road |
| City name | Gurgaon – 122002 |
| Phone No | 91 124 4739900 |
| Website | |
| Address | Regus, Level-12, Building 8C, DLF Cyber City, DLF Phase II, |
| City name | Gurgaon – 122002 |
| Phone No | 91 124 469 62 16 |
| Website | |
| Address | Plot No. 20, Sector – 18, Udyog Vihar Industrial Area |
| City name | Gurgaon-122001 |
| Phone No | 91- 124 2342001-10 |
| Website | |
| Address | 14th Floor, Tower D, Cyber Green, DLF City Phase-III |
| City name | Gurgaon 122002 |
| Phone No | 91-124-4313800 |
| Website | |
| Address | Plot No. 32, Sector 5, IMT Manesar, Haryana |
| City name | Gurgaon |
| Phone No | 91 124 418 7500 |
| Website | |
| Address | Gp-26, maruti industrial estate, Udyog vihar,sector-18, |
| City name | Gurgaon – 122015 |
| Phone No | 0124-3001243 |
| Website | |
| Address | Unitech Cyber Park, Tower C, 1st Floor, Sector 39, |
| City name | Gurgaon – 12200 |
| Phone No | 91 124 309 4000 |
| Website | |
| Address | 10th Floor, Tower C, Building #10, Phase – II, DLF Cyber City |
| City name | Gurgaon – 122002 |
| Phone No | 91-124-4960999 |
| Website | |
| Address | Plot# 92, Sector 32, Institutional Area, |
| City name | Gurgaon-122001 |
| Phone No | |
| Website | |
| Address | #447, Regus Business Centre, Level-4, Sector 53 , Augusta Point, |
| City name | Gurgaon-122002 |
| Phone No | 91 (11) 4060 1170 |
| Website | |
| Address | 510 JMD Pacific Square Near 32nd Milestone Sector 15, Part II |
| City name | Gurgaon – 122 001 Haryana |
| Phone No | |
| Website | |

- Share this post
Contract research organizations in Chennai
Contract research organizations in hyderabad, login to your account.
- Username/Email Address:
Forgot Password? | Sign Up
Reset Password
Already have an account? Login
Enter the username or e-mail you used in your profile. A password reset link will be sent to you by email.
Signup to your Account
- First Name *
- Last Name *
- Confirm Password *
- Organization Name
By clicking checkbox, you agree to our Terms and Conditions and Privacy Policy
Account Activation
Before you can login, you must activate your account with the code sent to your email address. If you did not receive this email, please check your junk/spam folder. Click here to resend the activation email. If you entered an incorrect email address, you will need to re-register with the correct email address.
- Your Email:
- Activation Code:
- Our Mission
- Synchron Brochure
- Therapeutic Experience
- Bio Statistics
- Bioanalytical Method
- Bioanalytical Expertise
- Pharmacokinetics & Biostatistics
- Data Management
- Medical Writing
- Quality Analysis
- Publications
- Presentations
- Advertisements
- Press Notes
- Be An Investigator
- Be A Volunteer
GET THE BEST CLINICAL RESEARCH SOLUTION IN INDIA
Quality Assurance
Synchron is a leading contract research organization in India, designed to meet the specific requirements of the clients in the field of :
- Clinical registration of new pharmaceutical products and medical devices, from phase I to phase IV, including Bioequivalence, Bioavailability, Pharmacokinetic / Pharmacodynamic studies, Statistical Analysis and Data Management strictly adhering to ICH and GCP guidelines.
- The company offers competitive and high quality clinical trial services to domestic and international pharmaceutical and Bio-Pharmaceutical companies.
- Phase I-IIA BA/BE
- Phase II-IV
- Bioanalysis
- Biostatistics & Data Management
- Quality Management
- Clinical Trials
| “Synchron House”, B/h. Mondeal Park, Nr. Gurudwara, S.G. Highway, Ahmedabad-380059 +91 -79 – 40235353, 26853419, 26840427, 26840348 +91-79-26853415 | |
| BIO -INNOVA AND SYNCHRON CO., LTD. | |
| 5 Webster Avenue, Summit, NJ 07901, USA +1 908 598 9636 +1 908 230 9373 +1 908 598 9638 |
- News & Events
Keeping pace with every evolving cell
Biosite is one of India’s leading Mid-Size CROs in the latest research, working with pharmaceutical companies and pan India.
Learn More…
Accelerating clinical trials with faster site selection and patient recruitment
Providing full and functional clinical research solutions to biotech, pharmaceutical and medical device industries worldwide.
THERAPEUTIC AREAS
In today’s R&D world there is no success without in-depth therapeutic expertise to support Trial Execution, Consulting and Placement solutions.
More than Just a living. View all our latest vacancies and learn more about a career at Biosite.
We are a multi-disciplinary laboratory committed to focussed and efficient drug discovery
Trusted partners.
Bangalore Office
Biosite Research, 2nd & 3rd Floor, Chandru Complex, RBI Layout, JP Nagar 7th Phase, J. P. Nagar, Bengaluru, Karnataka 560078
Contact Support
Contact Us: [email protected] Business Mail ID: [email protected] +91 080-35003479
© Copyright 2024 Biosite Research.
Designed by Webomindapps Pvt. Ltd. All Rights Reserved

- Search Search Search …
- Search Search …

Welcome to Xplora Clinical Research Services Pvt. Ltd. (XCRS)
We are keen to introduce ourselves as a fast growing Contract Research Organization (CRO) with inbuilt Site Management Organization (SMO), started in the year 2012. A team of clinical research professionals with more than 10 years experience in handling trial related activities in different phases are associated with it from its inception . As we are focused to improve the quality of clinical trial delivering on time and cost efficient trial outcomes to our client, we are promised to provide:
- Committed and helping our clients to meet their objectives and improve their trial designs and outcomes
- Providing a better quality of Investigators and research team for their products
- Helping in meet the targeted subject requirement on time and promised up to more than 25% of cost reduction
- Supporting our clients in recognizing their target and providing expertise for drug development processes
We provide employee excellence for our clients to reach their satisfactory requirements…
Biostatistics
Xplora has experienced professional of biostatistics
Clinical Programming
With intense and trained clinical practitioner
Clinical data Management
Xplora CRS provides top to bottom solution
Medical Writing
Medical writing has been added a core importance
Xplora Clinical Research Services
Xplora Clinical Research Services is a fastest growing clinical research organization specializing in the stipulation of management and conduction of clinical trials with its various aspects and procedures offering a fully integrated package with best quotation and service for its clients globally LEARN MORE
Expertise Services
Clinical trial logistics
Management Service
Clinical Data Management
XCRS provides solution for data management to create high quality and reliable data from clinical trial till the data base lock. XCRS Clinical data management team (CDM) is committed to provide fine data integrity with highest quality and strive the way for data management with eminence manner in compliance with industry standard
Site Management Facilities
XCRS completely understand the importance of sites and site based facilities and challenges faced by investigators in clinical research. We offer quality and potential investigators with well experienced in clinical research with awesome potential site for recruitment of subjects.
Medical writing has been added a core importance in field of medical science and clinical research. Increased number of clinical experiences and medical activities need to be communicated with its target and appropriate audience on time with an apposite and well-groomed deliverable. Our medical writing team closely works with our bio statisticians, Data management and clinical operations team in order to deliver quality reports and services on time with accuracy and cost effective in accordance with scientific standard.

- Chinese, Simplified
- Chinese, Traditional
- News & Resources
- Latest News
- Latest Articles
- Latest Reports
- Latest Webinars
- Upcoming Events
- Latest Case Studies
- Latest Whitepapers
- Latest FAQs
- Covid Notices
- Novotech Appoints Dr. Yooni Kim as a new executive position of Managing Director for APAC to Strengthen Global Expansion
- Novotech Honored with Frost & Sullivan’s 2024 Best Practices Company of the Year Award for Leadership in Biotech CRO Services
Novotech Earns Renewed Certification as an Employer of Choice for Gender Equality
- View All News
Pancreatic Cancer: Global Clinical Trial Landscape (2024)
Gastric cancer- global clinical trial landscape (2024).
- Oesophageal Cancer: Global Clinical Trial Landscape (2024)
- View All Reports
Empowering Patients & Challenges in Orphan Drug Development
Biostatistics in the development and evaluation of advanced therapies in china.
- Current Status and Prospects of Advanced Therapies in China
- View All Webinars
ESMO Congress 2024
- 12th Annual Outsourcing in Clinical Trials Southern California 2024
- View All Events
Examining a Phase 1 COVID 19 Clinical Trial
- Muscular Dystrophy – Multi-National and Multi-Site Trials Case Study
- Expert Consulting and Multi-Regional Clinical Trial (MRCT) Strategy Rescues Oncology Program
- View All Case Studies
Overview of Gastric Cancer: Global Clinical Trial Landscape
- Overview Of Pancreatic Cancer: Global Clinical Trials Landscape
- FAQs – RNA Therapies – Global Clinical Trials Landscape (2024)
- View All Faqs
RNA Therapy Clinical Trials Whitepaper – 2024
- Radiopharmaceutical Therapy Innovations & Global Clinical Trial Insights 2024
- Comprehensive Report on Antibody-Drug Conjugates Clinical Trials 2024
- View All Whitepapers
- South Korea and Australia on the rise as clinical research hubs
- Assessing the pros and cons of basing clinical trials in today’s European landscape
- Q&A with Judith Ng-Cashin, CRO Novotech's chief medical officer, on 2023 and beyond
- View All Article
Medical and Regulatory Consulting
Patient recruitment and site selection, early phase trials in australia, clinical operations and project management, site management organization (smo), biometrics and data management, virtual clinical trials, real world data, laboratory services, oncology cro services, pharmacometric services, drug development consulting, gmo solutions, liver disease cro services, infectious diseases and vaccines cro services, orphan and rare disease cro services, clinical and regulatory strategy, novotech appoints dr. yooni kim as a new executive position of managing director for apac to….
September 11, 2024, Boston, USA – Novotech, the leading global biotech Con...
Novotech Honored with Frost & Sullivan’s 2024 Best Practices Company of the Year Award for…
Boston, USA – September 10, 2024 – Novotech, the global full-service clinical Co...
Sydney, Australia – Sydney, Australia – Novotech, a global full-service clinical...
Explore cutting-edge pancreatic cancer research with Novotech CRO's disease report....
Discover the latest advancement in gastric cancer through Novotech CRO's in-depth disease analysis....
Join us for an insightful webinar on orphan drug development, where industry experts will share practical examples and key…
This webinar features a panel of industry experts discussing key statistical models and analysis within the therapy development…
Background:Phase I clinical studies are critical in the development of new therapies especially in the rapidly changing biotech...
Gastric cancer ranks as the fifth most diagnosed cancer and the leading cause of cancer-related deaths....
Explore the future of RNA therapeutics with Novotech's comprehensive whitepaper....
Covid-19 Notice important updates
Find content relevant to:.
Hong Kong, China
Barcelona, Spain
Novotech’s Medical and Regulatory Consulting team offers full range of pre-clinical, regulatory affairs support, medical and pharmacovigilance consulting services....
Novotech relies on years of experience, in-country knowledge and real-life big data to identify and propose the best-performing sites for...
Australia is a preferred destination for early phase trials because of simple and fast regulatory stream and lucrative R&D cash...
Novotech’s streamlined and integrated clinical trial services are delivered by a dedicated team of professionals with deep industry and therapeutic...
Discover the power of clinical excellence with Acrostar's SMO Division, a dedicated entity operating as part of Novotech....
Delivering accurate, high-quality and timely biostatistics in clinical trials services, including statistical planning, analysis and reporting....
How virtual clinical trials can offer patient retention and cost benefits compared to traditional trials....
Accelerating patient recruitment and drug development with real world data (RWD)...
Our bioanalytical services assist our customers in every stage of their molecule development....
The global Oncology landscape for Biotechnology companies...
Our team can assist in all clinical study phases and in study designs ranging from first in human, single ascending...
Novotech Drug Development Consulting is a full-service global product development and strategic regulatory group, providing comprehensive “lab to launch” program...
GMO Solutions: Novotech initiated the first national, privately owned, commercial Institutional Biosafety Committee (IBC) in Australia....
The global Liver Disease landscape for Biotechnology companies...
The global Infectious diseases and Vaccines CRO services landscape for Biotechnology companies...
The global Orphan and rare disease CRO services landscape for Biotechnology companies...
Clinical and Regulatory Strategy: Many biotechnology companies come to Australia to conduct early phase clinical trials and take advantage of...
Our workplace culture reflects the passion of our people, and we will support you to develop and achieve at all...
Novotech is a global full-service contract research organization (cro) providing clinical development services across all clinical trial phases and therapeutic..., there are many reasons people love working at novotech, but when you join it will be our open, inclusive, and..., we are committed to providing ongoing professional development training, a competitive bonus structure, a supportive work environment, variety in their..., our mission is to create career development opportunity for everyone...., we are committed to hiring ambitious and ethical professionals genuinely excited to be a part of the dynamic life sciences industry and who relish a challenge., search novotech, our company.
Founded in 1997, Novotech is a global full-service clinical Contract Research Organization (CRO) focused on partnering with biotech companies to accelerate the development of advanced and novel therapeutics at every phase.
Recognized for its industry-leading contributions, Novotech has received numerous prestigious awards, including the CRO Leadership Award 2023, and the Cell & Gene Therapy Clinical Trials Excellence 2023 award.
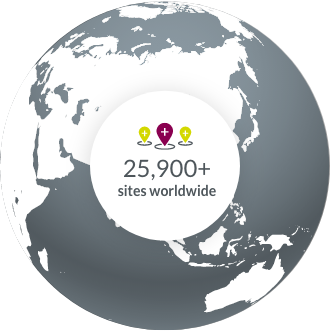
Novotech has experience in over 5,000 clinical projects, including Phase I to Phase IV clinical trials and bioequivalence studies. Novotech is positioned to serve biotech clients conducting clinical trials in Asia Pacific, the US and Europe. Novotech has over 3000 staff globally and 34 office locations.
Novotech provides clinical development services across all clinical trial including regulatory and drug development consulting, protocol design and writing, feasibility assessments, local ethics committee and regulatory submissions, clinical monitoring, project and vendor management, data management, statistical analysis, data visualization and analytics, DCTs, medical advisory and medical monitoring, pharmacovigilance, central lab services and clinical study report writing. We work to the highest quality standards, in line with ICH-GCP, global regulatory agencies and underpinned by robust processes in line with our ISO 9001 accredited Quality Management system.
Novotech has broad therapeutic experience with a particular focus on oncology/immuno-oncology, infectious disease/vaccines, neurology, and rare diseases.
As a biotech specialist, Novotech has extensive medical, scientific, regulatory, and operational expertise with advanced and novel therapeutics, across all therapeutic areas, including cell & gene therapies, precision medicine, RNA based vaccines and therapeutics, ADCs, microbiome, radiopharmaceuticals and more.
Our geographic presence, our depth of expertise and services, our operational infrastructure, and investments in technological advancements, combine to provide the perfect solution to accelerate global clinical development for biotech Sponsors.
Our Culture
Our values and our people is what makes Novotech a great place to work
Our work culture is driven by our values, every day.
Performance Passion
Making it happen, doing it well and owning the outcome
Uncompromised Integrity
We never hesitate to say or do the right thing
Open Engagement
How we work together recognizes the value of everyone involved through open communication
Unlocking Talent
We are all responsible for developing our people
Our mission is to help bring advances in therapeutic products to market efficiently by providing high quality clinical development services to our clients. We will do this by hiring and developing highly talented people, employing the best technologies in our operations, and by fostering a culture of open communication, teamwork and sharing of knowledge, experience and resources with our internal and external partners.
A horizontal organisational structure that encourages inter-country and inter-department collaboration and knowledge-sharing is paramount. We are proud of our inclusive culture and committed to always looking for ways to improve and remain a leader in workplace flexibility and gender equality. We are continually making progress
Board of Directors

Dr. Anand Tharmaratnam
Anand brings over 20 years of experience in global drug development and the CRO industry. He was previously the president of Asia Pacific at IQVIA. Anand is a medical doctor by training and graduated from the University College London, UK and trained in the anaesthesia.

Mr. Joel Thickins
Joel is a Partner at TPG Capital and leads the firm’s investment program in the ANZ region as Head of Australia and New Zealand. Joel has previously served on the board of Inghams Group Limited and Accolade Wines Holding Australia Pty Limited.
Before joining TPG, Joel was a director at CPE Capital (formerly known as CHAMP Private Equity) where he was responsible for the company’s activities in the industrial, agricultural services and consumer markets.

Dr. Fred Cohen
Fred Cohen is a Senior Advisor to TPG and was a partner and co-founder of TPG Biotech. Fred has served on the board of several biotech and pharmaceutical organizations including BioCryst Pharmaceuticals, Inc. (NASDAQ: BCRX) and CareDx, Inc. (NASDAQ: CDNA)

Mr. Vincent Wong
Vincent Wong is Partner with TPG Capital and is based in Melbourne, where he works in TPG’s Australian team. Vincent also serves on the board of other TPG investments such as Invocare, iNova, and Funlab. Prior to joining TPG in 2011, Vincent served in the corporate advisory division at Goldman Sachs Australia. Vincent graduated from the University of Melbourne with BCommerce and BLaws (Honors).

Ms. Cai Li (蔡俐)
Lydia Cai is a Managing Director with TPG Capital based in Beijing and leads TPG’s healthcare investments in the Greater China area. Prior to joining TPG in 2011, Lydia was an investment associate at HAO Capital, a China-focused early stage private equity fund focusing on growth-stage healthcare investments.

Dr. John Moller
John joined Novotech in January 2014 to lead the development of Novotech’s Asia operations. Prior to Novotech John was Managing Director of IVF Australia and Queensland Fertility Group and sat on the Board of Virtus Health Limited. John is a medical doctor by training.

Ms. Elaine Cheung (張綺玲)
Ms. Cheung is responsible for providing independent advice to the Board. She chairs the Audit Committee. Ms. Cheung has more than 30 years of corporate experience in the Asia-Pacific region. In January 1990, Ms. Cheung graduated from San Francisco State University with a bachelor of science degree, majoring in business administration.
Senior Management

Michael Stibilj
Michael has 25 years of experience in the CRO Industry. Prior to joining Novotech in 2017, Michael spent 15 years building and leading operations across Asia for Quintiles/IQVIA, and was senior advisor for TPG Capital.

Dr. Judith Ng-Cashin
Judith joined Novotech in 2023 bringing 20 years of international pharmaceutical, biotech and CRO experience, including multiple senior executive roles, including CMO and CSO roles. Judith, based in the US, is driving Novotech’s global multi-regional later-phase strategy as well as ensuring high quality delivery of medical services to our clients and patients.

Barry Murphy
Barry joined in July 2014 to lead the growth of Novotech’s Business Development team. Barry started his career as a pharmacist having graduated from the University of Dublin. He has held multiple contract management roles at ICON plc.

Rob Speedie
Rob was most recently a Managing Director in the TPG Capital Operations Team and lead a number of Strategic Initiatives across TPG’s portfolio, including time as the CPOO at Greencross. Prior to TPG, Rob was a Partner at PwC where he spent a total of 19 years. His roles at PWC included 10 years as a Transaction Services professional and 10 years in audit services across Australia, Asia and US. He worked in a range of industries including the pharmaceutical sector. Rob acted as Investigating Accountant and Signing Partner on multiple IPOs on the ASX, was a member of PwCs technical accounting issues escalation committee and has significant experience in financial due diligence.

Veronica Holloway
Veronica joined Novotech in May 2014 to formalise the legal function at Novotech. Prior to Novotech, Veronica held roles as Senior Legal Counsel at EY, Senior Solicitor at Clayton Utz and General Counsel (on secondment) at Novartis Pharmaceuticals Australia Pty Limited. Veronica holds a BA/LLB/LLM.

Angela Edwardson
Angela joined Novotech in November 2015 and is responsible for Novotech's Human Resources operations including strategy, organisational development, remuneration & reward and workplace health and safety. Angela is passionate about delivering Novotech's vision of being the leading CRO in the Asia Pacific region.

Andy worked at Covance, Inc., a global CRO, for more than 10 years in a number of executive positions. Most recently, he was General Manager of Covance's Central Laboratory Service, Asia Pacific, where he led a large multi-functional team of across multiple sites in APAC. Andy holds an MBA from the University of Chicago, an MS in Electrical Engineering from the Rose-Hulman Institute of Technology, and a BS in Electrical Engineering from Tsinghua University.

Iain Gordon
Iain, with over 20 years of experience in CRO management and corporate development in Europe and North America, joined EastHORN in 2013 as Chief Business Officer. He played a vital role in the company's growth. He has held senior management positions, including European Head and Global VP of Business Development, in the global CRO industry. In 2021, he succeeded Dr. Alan Wood as Executive Chairman, ensuring the company's growth continued apace. After the 2023 merger with Novotech, Iain became Managing Director of Novotech Europe and sits on their Executive Board. Based in the UK, he holds a degree in Cell Biology from the University of Glasgow.

Rick Farris
Rick joined Novotech in 2023 with over 22 years of clinical research experience and over 30 years in the life sciences. His clinical research career has been focused wholly on serving the unique needs and challenges of biotech clients. Rick has worked in executive leadership roles in 3 global CROs, leading operations teams and helping organizations build global project support capabilities.

Dr. Yooni Kim
Dr. Yooni Kim is Managing Director APAC at Novotech with over 25 years of clinical research experience and technical knowledge in CRO, Pharmaceutical and Academic Research.
With a Ph.D. in Preventive Medicine (Epidemiology) from the Seoul National University of Medicine, she started her career at GSK in 2004 and joined ICON Clinical Research (ex-PRA) in 2009 as Director of Operations.
Dr. Yooni worked with PRA Health Sciences for seven years, establishing the legal entity and service capabilities for PRA Korea. Since joining Novotech in 2016, she has provided exemplary service as the Executive Director Asia Operations before being appointed her current position of Managing Director APAC at Novotech.
Our Latest Awards


Standing in the gap between what is and what can be… Siro can help navigate and find answers that empowers lifesavers.
READ ON >
Rediscover efficiency with our accurate solutioning and customer-focused approach.
Rediscover dependablity with our market leading data management services.
Since decades, Siro continues to deliver in the global healthcare industry. See the highlights of our journey here.
END TO END CLINICAL RESEARCH ORGANIZATION
One stop shop for services ranging from medical writing and clinical operations, to data management and biostatistics.
GLOBAL CLIENTS
Long term strategic partnerships with large pharmaceutical companies from USA, Europe and Japan.
THERAPEUTIC EXPERTISE
World class therapeutic experts specializing across all niche therapeutic areas and indications.
SOLUTIONS SIMPLIFIED
Striving to provide a unique and unchallengeable solution to complex drug development processes.

Our Services

Trial Supplies
Follow the buzz
Powered by juicer.
- +91-7672005050
- [email protected]
Deep insight to clinical trials for the Indian market
An initiative of cliniexperts brings clinical trial services in india for local and global clinical trials.
Our clinical trial solutions are designed to address your specific needs and deliver a smooth development pathway
Expert Clinical Research Services For Product Lines

Clinical Trial Management Services
Efficiently launch your product with our customised services.
At CliniExperts Research Services, we focus on conducting high-quality trials in line with ethical and regulatory compliance. We aim to be your go-to Clinical Research Organization (CRO) by offering:
Whether you are a large corporate or an up-coming startup, our clinical trial solutions are designed to address your specific needs and deliver a smooth development pathway.
Therapeutics, Diagnostics & Wellness
Test products across diverse domains.
Our subject-matter experts guide you through each step of your product's launch so you don't have to worry about the hassle of regulatory approvals. Our range of services spans the following categories of products
Prerequisites for conducting clinical trials in India
Ethics committee registration.
We support the registration and re-registration of ethics committee (EC) to CDSCO via SUGAM portal.
Clinical Trial Registry India (CTRI)
To list your clinical trial on CTRI, we can assist you with CTRI Registration and CTRI submission over the CTRI online portal.
Credentials
Our success stories to tell.
FDCs must be based on convincing therapeutic rationalization and be carefully justified and clinically relevant. We helped our sponsor to prove the therapeutic rationale for various FDCs by conducting PIV clinical trials in India for gastroenterology, Gynaecology and Respiratory therapeutic areas
PMS/PSUR being conditioned for Market Authorization and licensing in India. The PSURs are to be submitted every six months for the first two years of the approval and for subsequent two years annually. We have conducted/conducting PMS studies for our sponsor as per “New Drugs and Clinical Trials Rule, 2019”. This has helped the sponsor to meet the prerequisites as per the regulations
To claim marketed drugs with modified or new claims including indication, route of administration, dosage and dosage form. A subsequent new drug (SNDs) approval required from CDSCO. Here, CliniExperts supported our client to conduct PIII, PIV and PMS studies as per CDSCO guidelines
Clinical trials for phytopharmaceutical drugs to be conducted as per applicable Rules and guidelines for new drugs. CliniExperts Research conducted three PIII clinical trials for three therapeutic conditions in India
CliniExperts has helped in conducting PII, PIII and PIV clinical trial in diversified therapeutic categories like Polycystic Ovary Syndrome (PCOS), Recurrent ascites with liver cirrhosis, Premenstrual Syndrome, Primary Dysmenorrhea and Alopecia Areata in children and adolescents
Most of the patients in intensive care unit (ICU) with impaired oral food consumption, present nutritional challenges which needs to be compensated using enteral tube feeding (ETF). We have successfully conducted safety and tolerance of nutritional supplements via tube feeding in hospitalized patients
Functional food supplements has grown exponentially in India and to prove their claims, feasibility and safety evaluation studies are required. CliniExperts Research has conducted various studies to prove their claims like Glycaemic Index evaluation of product on Diabetic and Pre-diabetic population and many more
Driving Clinical Excellence: Unveiling CliniExperts' Expertise
A seamless blend of scientific expertise, operational efficiency, and unparalleled client support
Expert Articles
Medical devices, best practices for designing medical device clinical investigations, biologicals |, diagnostic kits |, the drug development process – from conception to market · cliniexperts, consumer claims |, effective site management in clinical research – points to consider · cliniexperts, conducting clinical trial during covid-19 · cliniexperts, certifications and accreditations.
With these certifications, we demonstrate our dedication to quality, safety, and customer satisfaction.

- ICH GCP (De)
- ICH GCP (En)
- ICH GCP (Es)
- ICH GCP (Fr)
- ICH GCP (It)
- ICH GCP (Pt)
- ICH GCP (Ru)
- AUSTRALIA (NHMRC)
- JAPAN (PMDA)
- US Clinical Trials Registry
- EU Clinical Trials Registry
- Pharmaceutical Companies
- Clinical Research Labs
- Service Companies
- Clinical Research Events
- Publications
- Researchers
Top 15 Contract Research Organizations (CROs) in 2024

Post-pandemic, as drug development businesses recover and grow, CROs around the world continue to undergo profound transformation. As in previous years, 2023 will be all about mergers and acquisitions, with big players getting even bigger and smaller companies surviving thanks to niche advantages such as technological know-how or brilliant scientists among their employees.
The top 15 CROs globally would be in the following order based on 2023 revenue :
- LabCorp (Covance) , with 15.05 billion USD (2023) and 75000 employees ( Labcorp , 2024). In February 2015, Labcorp completes its $6 billion purchase of Covance, Inc., creating the world’s leading health care diagnostics company. The combination of Covance’s drug development leadership and Labcorp’s medical testing expertise builds the market leader in central laboratory and bioanalysis services. Labcorp’s clinical trials companies, Labcorp Clinical Trials and Tandem Laboratories, align under the Covance brand.
- IQVIA , with 14.85 billion USD (2023) and 86000 employees (IQVIA, 2022). IQVIA, formerly Quintiles and IMS Health, Inc., is an American Fortune 500 and S&P 500 multinational company serving the combined industries of health information technology and clinical research. IQVIA (NYSE:IQV) is a leading global provider of advanced analytics, technology solutions, and clinical research services to the life sciences industry. With approximately 86,000 employees, IQVIA conducts operations in more than 100 countries.
- ICON (PRA) , with 7.74 billion USD (2022) and 41160 employees (ICON, 2023). From a small team of 5 people in 1990, ICON now employs over 41,100 people. ICON has been recognised as one of the world’s leading Contract Research Organisations through a number of high-profile industry awards .
- Thermo Fisher Scientific (PPD) , with 7.02 billion USD (2023) and 35000 employees (Businesswire, 2023). Recognized as a global industry leader in accelerating promising medicines from early development through regulatory approval and market access, we serve pharma, biotech, medical device and government organizations with custom-tailored solutions, including full-service partnerships and functional service partnerships. As a strategic partner in clinical development and analytical services, we apply cutting edge technologies, therapeutic expertise and a firm commitment to quality to help customers deliver life-changing therapies.
- Lonza - 6.5 billion USD, 17896 employees. Though being a contract development and manufacturing organization (CDMO), rather than a CRO, takes the 10th position with the 5.9 billion USD in sales and over 17500 employees worldwide (2023), offering “proprietary line of in silico and in vitro services for manufacturability, immunogenicity, potency assessment, humanization and protein engineering” ( https://pharma.lonza.com , 2022). Founded in 1897 in the Swiss Alps, today, Lonza operates across five continents. With approximately 17,500 full-time employees, it comprises high-performing teams and individual talent that make a meaningful difference to our own business, as well as to the communities in which we operate. The business benefits from global supply chains, but we have worked to maintain the agility to address marketplace needs on a local level. CDMOs become reliable partners for Pharma companies as their “services reduce R&D costs while improving productivity and are essential to providing safe and effective treatment to patients” ( https://pharma.lonza.com , 2022.
- Wuxi AppTec with 5.8 billion USD revenue in 2021 and 44360 employees (Companiesmarketcap, 2022). As a global company with operations across Asia, Europe, and North America, WuXi AppTec provides a broad portfolio of R&D and manufacturing services that enable the pharmaceutical and healthcare industry around the world to advance discoveries and deliver groundbreaking treatments to patients. Through its unique business models, WuXi AppTec’s integrated, end-to-end services include chemistry drug CRDMO (Contract Research, Development and Manufacturing Organization), biology discovery, preclinical testing and clinical research services, and cell and gene therapies CTDMO (Contract Testing, Development and Manufacturing Organization), helping customers improve the productivity of advancing healthcare products through cost-effective and efficient solutions. WuXi AppTec received AA ESG rating from MSCI in 2023 and its open-access platform is enabling more than 6,000 customers from over 30 countries to improve the health of those in need – and to realize the vision that "every drug can be made and every disease can be treated."
Syneos Health® is a leading fully integrated biopharmaceutical solutions organization built to accelerate customer success. We translate unique clinical, medical affairs and commercial insights into outcomes to address modern market realities.
We bring together a talented team of professionals, who work across more than 110 countries, with a deep understanding of patient and physician behaviors and market dynamics. Together we share insights, use the latest technologies and apply advanced business practices to speed our customers’ delivery of important therapies to patients.
Cell and Gene Therapy CDMO Solutions has supported the development of 11 FDA approved cell and gene therapies and have conducted more than 900 studies in this field over the past year. Unsurpassed end-to-end offering results in enhanced access to scientific and regulatory expertise via multidisciplinary bench of experts to help you problem solve.
Parexel (EQT Private Equity and Goldman Sachs Asset Management) with 3.8 billion USD in 2022 and 21000 employees (Parexel, 2023). Parexel is among the world’s largest clinical research organizations (CROs), providing the full range of Phase I to IV clinical development services to help lifesaving treatments reach patients faster. Leveraging the breadth of our clinical, regulatory, and therapeutic expertise, our team of more than 21,000 global professionals works in partnership with biopharmaceutical leaders, emerging innovators, and sites to design and deliver clinical trials with patients in mind, increasing access and participation to make clinical research a care option for anyone, anywhere.
- Medpace Holdings, Inc. with 1.135 billion USD revenue in 2021 and 5400 employees (Medpace, 2023). Integrating core clinical trial services delivers efficient and streamlined execution and higher quality results. Through its wholly-owned subsidiaries, Medpace offers clinical pharmacology, as well as supporting laboratory services including central labs, bioanalytical lab, ECG core lab, and imaging core labs.
Worldwide Clinical Trials – 0.6 billion USD , 3000 employees, 60 countries.
Worldwide Clinical Trials started out more than 30 years ago as a small science team with a big goal: To always provide authentic, personalized attention to our partners. And as the industry has grown, CROs have merged, and trials have grown increasingly complex, we haven’t forgotten what truly matters: People.
With more than 3,000 employees in more than 60 countries around the world, we’re still staying true to those roots. Our team is dedicated to staying accessible, flexible, and solution-focused to ensure our partners not only have the best possible outcomes – but they also know their Worldwide team is only a call away.
Allucent (CATO SMS and Pharm-Olam) – 0.36 billion USD, 1200+ employees .
Allucent originated with CATO SMS, itself created by the merger of Cato Research and SMS-oncology in 2019. Cato Research, founded in 1988, was known for its ability to design and execute successful development strategies and guide creative new products through the regulatory process.
Previous acquisitions by CATO SMS included Array Biostatistics, a full-service biostatistical and statistical programming CRO, and Nuventra Pharma Sciences, one of the industry’s leading providers of clinical pharmacology science and services. With these acquisitions, CATO SMS expanded its services to offer biostatistical consulting, analysis, programming, and cutting-edge modeling and simulation techniques to inform clinical trial designs and predict trial outcomes.
In early 2022, CATO SMS merged with Pharm-Olam, a global clinical research organization delivering clinical trial services to organizations around the world.
KCR – 0.15 billion USD, 700 employees
KCR is a clinical development solutions provider creating value for biotechnology and pharmaceutical organizations.
Founded in 1997, our expert teams support clients with full-service clinical development capabilities across three main areas: Trial Execution, Consulting, and Placement. KCR operates across North America, Europe, and Australia, with main office locations in Boston, US, Berlin, Germany, and Warsaw, Poland.
Our geographical coverage across 25+ countries, cutting-edge technical capabilities and tailored offerings allow for the optimized delivery of solutions to develop life-changing therapies. KCR offers access to an estimated population of 1.1 billion people.
Advanced Clinical – 0.1 billion USD , 1100+ employees
Today, Advanced Clinical has grown organically, with coverage across North America, Eastern Europe, Western Europe and Asia-Pacific, providing contract research organization (CRO), FSP and strategic resourcing solutions. A research services and strategic resourcing organization committed to providing a better clinical experience by delivering comprehensive Contract Research Organization (CRO), Functional Service Provider (FSP) and Strategic Resourcing services that optimize effectiveness for both patients and sponsors throughout the clinical research journey.
Using our decades of experience, we strive to improve the lives of everyone touched by clinical research and help our clients achieve better outcomes through candid conversations, foresight, resilience and innovative solutions.
CTI Clinical Trial and Consulting Services – 0.08 billion USD, 750 employees . CTI has grown consistently and significantly, becoming a global organization with associates in more than 60 countries across the world. We have worked on more than 10,000 projects, worked on every continent except Antarctica, and have contributed to more than 150 new drug and device approvals through global regulatory agencies such as the FDA, EMA, and others. Currently, we work with approximately 250 pharmaceutical and emerging biotechnology and medical device companies.
Among recent reasons for mergers and the whole new approach to conducting clinical research, apart from pursuit of new clients, talents, expertise, products, and markets, there have been notorious coronavirus crisis and military conflicts worldwide. We faced the advent of globalization, decentralization, remote monitoring, and personalized approach to drugs R&D. But we are now looking into the era of technology in clinical research. Processes that allow remote audits and monitoring, previously considered too risky, are now risk mitigators (Bahls, Christine, 2021), at least from that point of view. Clinical trial participants enrollment, and even participation itself, constantly moves into tech dimension.
Only few among the leading CROs escaped M&As for now, but this seems to be the matter of time. Savlovschi – Wicks, Theodora (2022) estimated that “in 2021, the global CRO market is expected to reach an impressive US$88 billion by 2028”. According to IQVIA , only in biopharma sector the total transaction value grew by 70% to reach 152 billion USD by the end of 2023, featuring prominent $43Bn Pfizer-Seagen deal, and other considerable M&As for BMS-Karuna ($14Bn), Merck-Prometheus ($10.2Bn), AbbVie-Immunogen ($10.1Bn), AbbVie-Cerevel ($8.7Bn), Biogen-Reata ($7.3Bn) and Roche-Telavant ($7.1Bn).
Among the CRO businesses the following M&As took place lately:
- Charles River Labs acquires MPI Research (2018), Citoxlab (2019), HemaCare (2020) and Cognate BioServices (2021)
- IQVIA (formerly Quintiles and IMS Health) acquired Clintec and 40% of Q2 Solutions (2021)
- ICON purchased PRA Health Services (2021)
- Thermo Fisher Scientific bought PPD (2021)
- Labcorp purchased Covance and Chiltern
- Inotiv acquired Envigo (2021)
- INC Research and inVentiv Health merged to later become Syneos Health
- Parexel become the property of EQT Private Equity and Goldman Sachs Asset Management
- CATO SMS and Pharm-Olam merged in 2022
- Triley Bidco acquired Clinigen (2022)
The article by Nataliia Vietchinkina, MS in Clinical Research Administration
University of Liverpool.
References:
- Christine Bahls (2021) ‘The Post-Pandemic CRO Landscape’, Applied Clinical Trials, Applied Clinical Trials-09-01-2021, Volume 30, Issue 9 [online] Available from: https://www.appliedclinicaltrialsonline.com/view/the-post-pandemic-cro-landscape
- Savlovschi – Wicks, Theodora (2022) ‘Top 10 CROs to watch in 2022’ [online]. Available from: https://www.proclinical.com/blogs/2022-3/top-10-cros-to-watch-in-2022
- Labcorp (2022) Labcorp Announces 2021 Fourth Quarter and Full-Year Results, 10 February 2022 [online]. Available from” https://ir.labcorp.com/news-releases/news-release-details/labcorp-announces-2021-fourth-quarter-and-full-year-results#:~:text=Revenue%20was%20%2416.12%20billion%20%2C%20an,by%20divestitures%20of%20(0.1%25)
- IQVIA (2022) IQVIA Reports Fourth-Quarter and Full-Year 2021 Results; Raises Full-Year 2022 Profit Guidance, 15 February 2022 [online]. Available from: https://ir.iqvia.com/press-releases/press-release-details/2022/IQVIA-Reports-Fourth-Quarter-and-Full-Year-2021-Results-Raises-Full-Year-2022-Profit-Guidance/default.aspx#:~:text=Revenue%20of%20%2413%2C874%20million%20for,12.4%20percent%20at%20constant%20currency
- ICON (2022) ICON Reports Fourth Quarter and Full Year 2021 Results, 2022 [online]. Available from: https://www.iconplc.com/news-events/press-releases/icon-reports-fourth-quarter-and-full-year-2021-results/
- Businesswire (2021) ‘PPD Reports Fourth Quarter and Full Year 2020 Results’, 23 February 2021 [online]. Available from: https://www.businesswire.com/news/home/20210223006094/en/PPD-Reports-Fourth-Quarter-and-Full-Year-2020-Results
- Macrotrends (2022) ‘Syneos Health Revenue 2012-2021 | SYNH’ [online]. Available from: https://www.macrotrends.net/stocks/charts/SYNH/syneos-health/revenue
- https://companiesmarketcap.com/ (2022) ‘Revenue for Charles River Laboratories (CRL)’ [online]. Available from: https://companiesmarketcap.com/charles-river-laboratories/revenue/
- Vinluan, Frank (2021) ‘CRO Parexel changes private equity hands again, this time for $8.5B’ [online]. Available from: https://medcitynews.com/2021/07/cro-parexel-changes-private-equity-hands-again-this-time-for-8-5b/
- Medpace (2021) ‘Medpace Holdings, Inc. Reports Third Quarter 2021 Results’ [online]. Available from: https://investor.medpace.com/news-releases/news-release-details/medpace-holdings-inc-reports-third-quarter-2021-results#:~:text=The%20Company%20forecasts%202021%20revenue,%24176.0%20million%20to%20%24180.0%20million%20 .
- Companiesmarketcap (2022) ‘Revenue for WuXi AppTec (2359.HK) Revenue in 2021 (TTM)’ [online]. Available from: https://companiesmarketcap.com/wuxi-apptec/revenue/
- https://pharma.lonza.com (2022) ‘Design, assess and optimize for clinical success’ [online]. Available from: https://pharma.lonza.com/offerings/early-development-services .
Clinical Research News
Upcoming clinical trials.
- Harvard Pilgrim Health Care Stanford University Not yet recruiting Eco-labels on Restaurant Menus Dietary Habits | Food Preferences | Healthy Diet | Food Selection | Nutrition United States
- The University of New South Wales Evolution Health Pty Ltd Not yet recruiting LaCE (Lactobacillus Paracasei LPB27 On Early Childhood Eczema) Allergy | Eczema Australia
- Nanfang Hospital, Southern Medical University Recruiting A Multicenter Study Based on Multi-omics Analysis to Predict the Early Prognosis and Recurrence Risk of Acute Ischemic Stroke Ischemic Stroke, Acute China
- Kessler Foundation Not yet recruiting NEUROBALANCE Training to Improve Postural Control in Individuals with Traumatic Brain Injury Brain Injuries, Traumatic | Traumatic Brain Injury | TBI | Diffuse Axonal Brain Injury
- Leiden University Medical Center Not yet recruiting Calibration of Esophageal Balloon Catheter in Spontaneous and Mandatory Mechanical Ventilation. (PESCA) Critical Illness | Intensive Care Unit | Mechanical Ventilation
- VULM s.r.o. Recruiting The Purpose of This Study is to Assess Clinical Efficacy and Safety of Berovenal® Intended to Promote Treatment and to Expedite Chronic Diabetic Foot Ulcer Healing Diabetic Foot Ulcer
- Assiut University Not yet recruiting Evaluation of Serum Gasdermin D Level As a Potential Biomarker of Disease Activity in Vitiligo Patients Serum Gasdermin D in Vitiligo
- Universidad de Granada Not yet recruiting Understanding Cerebral Blood Flow Dynamics for Alzheimer's Disease Prevention Through Exercise (flADex) Healthy Aging | Blood Flow | Cognitive Function 1, Social | Cerebrovascular Circulation
- Riphah International University Recruiting Effects of Sub Occipital Muscle Inhibition Technique on Hamstring Flexibility in Post-laminectomy Patients. Hamstring Tightness Pakistan
- Cairo University Not yet recruiting Analgesic Efficacy of External Oblique Intercostal Block vs Subcostal Transversus Abdominis Plane Block in Open Surgical Nephrectomy Analgesic Efficacy | Nephrectomy | Open | Subcostal Transversus Abdominis Plane Block | External Oblique Intercostal Block Egypt
- Riphah International University Recruiting Effect of Low-Volume HIT vs. MIC Exercise in Individuals With Increased BMI Overweight and Obesity Pakistan
- Riphah International University Not yet recruiting Short Foot Exercises and Anti-pronation Taping on Low Back Pain Associated With Hyper Pronation Low Back Pain Pakistan
Clinical Research Jobs
Sponsors and Collaborators
- Assiut University
- GlaxoSmithKline
- National Cancer Institute (NCI)
- Cairo University
- AstraZeneca
- Assistance Publique - Hôpitaux de Paris
- Mayo Clinic
- M.D. Anderson Cancer Center
- Novartis Pharmaceuticals
- National Institute of Allergy and Infectious Diseases (NIAID)
- Massachusetts General Hospital
- National Taiwan University Hospital
- Boehringer Ingelheim
- Hoffmann-La Roche
- Merck Sharp & Dohme LLC
- Stanford University
- University of California, San Francisco
- Eli Lilly and Company
- Duke University
- View Full List
Medical Conditions
- Breast Cancer
- Hypertension
- Prostate Cancer
- HIV Infections
- Coronary Artery Disease
- Heart Failure
- Diabetes Mellitus, Type 2
- Colorectal Cancer
- Lung Cancer
- Schizophrenia
- Cardiovascular Diseases
- Atrial Fibrillation
Drug Interventions
- Cyclophosphamide
- Carboplatin
- Dexamethasone
- Gemcitabine
- Capecitabine
- Bevacizumab
- Pembrolizumab
- Methotrexate
- Oxaliplatin
- Dexmedetomidine
- Fludarabine
Processing...
- Appointment
First JCI & NABH Accredited Multispeciality Health Care Hospital in Gurgaon
- Hospital Overview
- Vision, Mission & Core Values
- Board of Directors
- Management Team
- Awards and Accreditations
- Medical Technology at Artemis
- Quality & Safety
- Bespoke Services
- Artemis Health Sciences Foundation
- Billing & Insurance
- Emotional & Support Programs
- Executive Health Services
- International Patient Services
- Medical Interpreter Services
- Medical Records
- Mind Body Medicine
- Patient and Family Advisory
- Patient Advocacy
- Patient Portal
- Pre-Admission Testing
- Smoking Cessation Programs
- General Information
- Cost and Insurance
- Accommodation
- International patients
- Request an Appointment
- Our doctors
- Appointments
- Visiting Times
- Virtual Tour
- Hospital Roadmap
- Contacting Patients
- Allergy and Immunology
- Blood (Haematology)
- Bones, Muscles and Joints (Orthopaedics)
- Brain, Spine and Nervous System
- Cancer (Oncology)
- Circulatory (Vascular)
- Dermatology
- Digestive (Gastroenterology)
- Endocrine, Diabetes And Metabolism
- Heart (Cardiology)
- Infectious Diseases
- Kidneys (Renal)
- Lung (Pulmonary)
- Mouth, Teeth And Face (Oral And Maxillofacial)
- Obstetrics And Gynaecology
- Rheumatology (Arthritis And Joint Disease)
- Specialities
- Artemis Corporate Social Responsibility
- Solace Home Care
- Apply Online
- Employee Speak
- HR Vision & Philosophy
- Life at Artemis
| You are here: | › | Research |
- Enhancing the quality of patient care and treatment through a research facility that strives to provide evidence based solutions and establishes strong linkages within the healthcare ecosystem.
- Fostering and promoting a research culture to enhance the image and academic foot print of the organization.
- Undertaking intramural and extramural research projects and collaboration with national and international scientific organizations leading up to peer reviewed, high impact publications.
- To provide inter-disciplinary training for scholars as well as supporting education through PhD and various training programs.
- Encouraging faculty from various clinical departments to be involved in research through Faculty Development Program.
Quick links:
- Find a doctor
- Patient visitor information
Centres of Excellence
- Artemis Emergency & Trauma Centre
- Artemis Heart Centre
- Artemis Cancer Centre
- Artemis Neurosciences Centre
- Artemis Joint Replacement & Orthopaedics Centre
- Artemis Minimally Invasive & Bariatric Surgery Centre
- Artemis Transplant Centre (Liver, Kidney, Cornea & Bone Marrow Transplant)
- Artemis Women & Child Centre
- Artemis Pulmonology & Critical Care Centre
- Artemis Gastrosciences Centre
- Artemis Cosmetology & Plastic Surgery Centre
- Artemis Pain Medicine & Palliative care
For Patients:
For visitors:, multimedia:.
- Public Information
- Media Coverage
- Health Campaigns
- Articles by Doctors
Why Artemis:

Artemis Hospitals
Sector 51, gurugram 122001, haryana, india, emergency phone no.:, +91-124 - 4588 888, artemis phone no.:, +91-124 - 4511 111, international patient queries:, +91 9711209940, [email protected].

- Corporate Info
- Terms Of Use
- Privacy Policy
- Another action
- Something else here
What are you looking for?
You want a partner that is as committed to your success as you are., latest news.
Syneos Health Leaders Recognized as PM360 ELITE 100 Award Recipients
Syneos Health Releases 2024 Dealmakers' Intentions Survey
Syneos Health Releases 2024 Health Trends
Syneos Health Appoints Colin Shannon as Chief Executive Officer
Syneos Health Closes Transaction with Private Investment Firms
Syneos Health Names Terttu Haring President, Clinical Sites & Patients
At Syneos Health you’ll find:
A partner with purpose.
When we partner, your goals become our goals. We listen and bring specialized experts together when and where you need them to support your teams and immediately add value increasing confidence at each step on your journey.
Therapeutic Depth
Our commitment to the success of our customers starts with how we craft individual project teams. Each team includes specialized experts suited to meet your unique needs, delivering deep insights to accelerate your therapies to market.
Insights-Based Solutions
Our integrated solutions span the entire clinical-to-commercial spectrum and are based on AI- and tech-enabled insights designed to improve performance, reduce risk, and speed much-needed therapies to patients worldwide.
FEATURED SOLUTIONS
Our therapeutic expertise in oncology and hematology runs deep and wide. Our unique lab-to-life service offering extends from preclinical and translational science to clinical development and regulatory to full commercialization and marketing capabilities.
We’re passionate about collaborating for a cure.
Predictive Commercial Solutions
Blending a deep understanding of human behavior and the latest technology with our hard-earned industry knowledge and insights gained from supporting the commercialization of more products than any other partner, we deliver predictive solutions that reduce risk, accelerate commercial performance, and fulfill our industry’s promise to patients.
Seeing new market realities and pathways to success.
Ophthalmology
Since 2010, Syneos Health has executed 290+ ophthalmology projects, including 35 Wet AMD studies, and developed and commercialized 48 approved eye care products, making us a leading ophthalmology contract research organization providing clinical development and commercialization services for drugs, devices and diagnostics in vision care.
SYNEOS HEALTH G.K. サイネオス・ヘルス合同会社
職務執行者 スティファン グトウ 職務執行者 ジュリー・マリー・ロビンソン サイネオス・ヘルス合同会社は、サイネオス・ヘルス・コマーシャル株式会社とサイネオス・ヘルス・クリニカル株式会社の持ち株会社です。2社の株式保有ならびに日本およびアジア太平洋地域のサイネオス・ヘルス グループ各社の事業支援を行います
SYNEOS HEALTH COMMERCIAL K.K. サイネオス・ヘルス・コマーシャル株式会社
代表取締役 セオドア・シーチャオ・ウォン サイネオス・ヘルス合同会社は、ササイネオス・ヘルス・コマーシャル株式会社は今回の事業再編のため新たに設立され、旧インヴェンティヴ・ヘルス・ジャパン合同会社よりコマーシャル事業を継承しました。この新会社が日本においてコマーシャル事業を展開いたします。
SYNEOS HEALTH CLINICAL K.K. サイネオス・ヘルス・クリニカル株式会社
代表取締役 スティファン グトウ 旧INC Research Japan株式会社は、旧インヴェンティヴ・ヘルス・ジャパン合同会社より、クリニカル事業を承継、統合のうえ、サイネオス・ヘルス・クリニカル株式会社と社名を変更いたしました。日本においてクリニカル事業を展開いたします。
より迅速なプロセス、より深い洞察、より効率的なシステム、より実用的なリアルワールド・エビデンス、およびそれらに対する理解。これらは、サイネオス・ヘルスと「バイオ医薬品ビジネスの加速化」が、日々複雑化を増し、競争が激化する医薬品市場にもたらすメリットの一部です。

SYNEOS HEALTH INSIGHTS HUB
Your source for future-focused, actionable insights to help biopharmaceutical companies better execute and succeed in a constantly evolving environment.

Genetic Medicine Leadership: Pioneering Science That Rewards Innovation

Artificial Intelligence and Alzheimer’s: Transforming Patient Screening with Predictive Models

WEBINAR | 2024 Health Trends: The Reinvention of Life Science

Driving Impact: Key Themes for Advancing Medical Affairs in the Evolving Pharma Landscape
Syneos health careers, work here matters everywhere.
Each day, you’ll be reminded about why you chose a career in healthcare. The work is challenging. The pace is exhilarating. The people – your colleagues and our customers – are inspiring.

Therapeutic Expertise
Participate

Explore end-to-end solutions throughout development — from portfolio optimization and regulatory strategy, to Phase I-IV clinical trials, market access planning, and more.
- Portfolio management and asset valuation
- Early development and innovation
- Integrated clinical development
- Approval and access
- Value substantiation lifecycle management
HOW WE DO IT
- Delivery models
- Operational excellence
- Medical affairs
- Building patient insights into assets, profile and claims
- Portfolio optimization
- Asset valuation and indication prioritization
- Early evidence review
- Model-based drug development
- Integrated development strategy and planning
- Phase I Clinical Trials
- Proof of Concept Studies: Phase IB-IIA
- Patient Engagement Strategy and Enrollment Solutions
- Patient Inclusion
- Site Alliance Network and KOL Engagement
- Protocol Optimization
- Regulatory Strategy
- Market Access Strategy and Delivery
- Biomarker and Genomic Medicine Strategy
- Clinical Trial Supply & Logistics
- Medical Communications
- Phase IIB-IV Clinical Trials
- Operationalize post-trial access with a proven platform approach
- Protocol-Driven, Customized Site Solution Strategy
- Regulatory Strategy, Submissions, Compliance, and Outsourcing
- Clinical Development Technology Optimization
- Real World Evidence
- Global Regulatory Submissions and Outsourcing
- Compliance and Risk Management
- Real-World Evidence, Market Access Strategy and Planning
- Regulatory Compliance, Drug Safety and Pharmacovigilance
- Lifecycle Optimization
- Gain an advantage through FSP
- Leveraging AI and digital in clinical development

Parexel Biotech provides the end-to-end capabilities you’ll need to succeed. We're fast, flexible, and dedicated to impacting patient lives.

Utilize our expertise across therapeutic areas, combining innovative trial designs, leading clinical and regulatory expertise, global reach, and a passion for changing patient lives.
Neuroscience
- General medicine
- Infectious disease & vaccines
- Inflammation & immunology
Cross-Therapeutic Expertise
- Cell & gene therapies
- Rare diseases

Our experts help you stay at the forefront of the industry - and ahead of change.
New Medicines, Novel Insights
- Advancing precision oncology
- Advancing rare disease drug development
- Accelerating development of cell and gene therapies
- Achieving patient-guided drug development
Discussions on Diversity
- Chapter 1 Bridging the Gap
- Chapter 2 Beyond the Binary
The Regulatory Navigator
- Gene therapy: are high costs and manufacturing complexities impeding progress?
- New FDA draft guidance: Implications for simplifying interchangeability for biosimilars in the US
- Key implications for nonclinical development: FDA guidance on human gene therapy products incorporating human genome editing
Latest Report

New Medicines, Novel Insights: Advancing precision oncology

Thinking about joining a clinical trial? Learn the drug development process, what it’s like to participate, how to find a trial, and answers to frequently asked questions.
INTERESTED IN PARTICIPATING?
- Healthy Volunteers
- Patient Volunteers
- Patient Advocacy Groups
HEAR FROM REAL PATIENTS
- Patient Stories
TRIAL SITES

Want to collaborate with us to offer clinical trials at your site? We would welcome the opportunity to discuss.
We are one of the largest CROs in the world, speeding life-changing medicine to market by engaging patients With Heart ™. Learn about who we are, what we do, and what we believe.
- Leadership team
- Global reach
- Our DE&I strategy
- Compliance, tax & privacy
- Meet us at an event
- Board of Directors
- Our ESG strategy
- Compliance & Ethics
- Tax Strategy
- Privacy Policy
- Cookies and other web tracking
- Supplier Data Privacy Requirements
- Terms of Use
- French Gender Pay Data
- UK Gender Pay Data
- UK Modern Slavery Act Statement
- Data Privacy Framework
- Supplier Diversity Policy
What can we help you find today?
The perspectives and opinions expressed in this material represent those of the patient advocate only and should not be considered a solicitation, promotion or advertisement for any services of Parexel, or any drugs or therapies, including those under development. Participating in clinical trials for investigational medicines offers patients potential benefits, such as access to cutting-edge treatments and expert medical care, while contributing to medical research. However, risks may include side effects, unpredictable outcomes, and time commitment. Careful assessment of these factors helps patients make informed decisions. The content of this material, including graphics, images and text, is provided for informational purposes only and does not constitute medical advice, diagnosis or treatment. Please consult your healthcare professional for medical advice. The patient advocate has provided their consent for the use and distribution of this content.
Patient Story
- Inflammation & Immunology
Rare Diseases
- Cell & Gene Therapy
Tina had lost her father to Crohn's disease. Now she faced her own diagnosis.
Then came the diagnosis. Finding out she had Crohn’s disease, one of two IBD syndromes that affect 10 million people worldwide, wasn’t a surprise considering her family’s history with the disease. But it derailed her life.
Tina was in her early 20s. She lived in New York City and was building a promising future for herself in finance.
She spent the next 10 years trying countless medications and underwent 20 surgeries. nothing worked., then, finally, she enrolled in a clinical trial., within months, she was in remission—and has been ever since., today, she’s a patient advocate, sharing her experiences to help keep our clinical trials focused on the needs of patients like her., lives can change when you design inflammation and immunology trials with agility, precision, and care..
- Keep up with urgent clinical demands
- Utilize the right experts, with deep I&I experience
- Help participants with chronic conditions feel safe and cared for
- Get seamless, end-to-end support across indications.
What we do, we do
Our Experts
Our inflammation and immunology specialists collaborate to get your treatment to patients faster.
MORE EXPERTS
Bharti Arya
Executive Director, ...
Michelle Pietzak, M.D.
Global TA Section He...

Mwango Kashoki, M.D., M.P.H.
Senior Vice President, Global Head of Regulatory Strategy
Mwango brings over 20 years of clinical and regulatory drug development experience to her role at Parexel, including more than 16 years at the U.S. Food and Drug Administration (FDA). She provides strategic and technical guidance on various regulatory and clinical aspects of drug development, leveraging her expertise in clinical review, pharmacovigilance and post-approval safety requirements across multiple therapeutic areas. Mwango is a critical asset for our customers, helping them navigate the changing regulatory environment on their development journeys to deliver important new treatments to patients in need across the globe.
Our I&I team is 13,100+ strong, with experience across indications, to match the needs of your trial.

Our diverse project experiences and expertise in patient recruitment and engagement result in fast, efficient trials.

Clinical trial sites around the world allow us to accelerate study startups.
- North America
- South America
- Middle East & Africa
Innovative trial designs allow us to optimize your trials for maximum impact.
And our patient-first approach results in deeper, more relevant insights for trial design and execution.
BENEFITS MAY INCLUDE

What can we do to help you change patient lives?
See I&I capabilities Visit all therapeutic areas Explore I&I careers
Discover other patient stories
Sara's only symptom was fatigue. But after some routine tests, she was diagnosed with breast cancer.
When Austin was 3 years old, his parents realized something was wrong. Multiple falls, concussions, and a broken arm led to a diagnosis.
CELL & GENE THERAPY
One night, while watching TV with her husband, Robyn felt a lump in her neck. As a doctor, she knew the risk.
While running a triathlon, Andrea stumbled and realized something was wrong. She thought it was just an injury. But it was much more.
We focus on patients, because they inspire us to deliver better trials, faster than ever. So we can make a difference for more patients like Tina.
Who we are,, parexel is proudly among the world’s largest clinical research organizations.
A dedicated CRO providing the full range of Phase I to IV clinical development services and leveraging the breadth of our clinical, regulatory and therapeutic expertise , our team of more than 21,000 global professionals works in partnership with biopharmaceutical leaders and sites to design and deliver clinical trials with patients in mind , to make clinical research a care option for anyone, anywhere.
- gurgaon News
Gurugram University to provide advanced certificate course in Clinical Research

Visual Stories

anovus Institute of Clinical Research
Welcome to anovus Institute of Clinical Research, where we offer top-notch job training services in the field of Health & Wellness. Our institute has been established in 2009 and has since then trained numerous professionals in the area of clinical research. With a team of dedicated and experienced trainers, we provide comprehensive training in pharmacovigilance, clinical research, and online job training. Our training programs are designed t ... less

Compilation Avenue (Capilt Services Pvt. Ltd.)
Compilation Avenue ( A venture of CAPILT Services Pvt. Ltd.) is a complete and secure solution for your business process outsourcing needs. Whether it is to compile and evaluate your massive data rapidly or you want to have multi-channel touch points with your customers. Our outcome-driven solutions produce immediate lift, enriched and valued results. With our Data Management Services you can manage & process your business ... less
G.B.K. Bio Sciences
National brain research centre, anahat clinics, apothecaries clinical research, institute for human development, you may also interested.
19580+ Service Professionals
4600+ Service Professionals
12280+ Service Professionals
15770+ Service Professionals
18640+ Service Professionals
7240+ Service Professionals
5390+ Service Professionals
10200+ Service Professionals
31530+ Service Professionals
15760+ Service Professionals

COMMENTS
CRO Name: Accenture Address: 1st floor, Tower B & C, Building No. 8, DLF Cyber City Phase II: City name: Gurgaon - 122002: Phone No +91 124 452 0000: Website
High disease population load. The clinical activity grew by over 40% in 2018 on the back of regulatory pathway improvements. About 1,800 investigators have participated on a minimum of 5 studies. India is the world's second most populated country with 1.3 billion people. Over the past decade, India has implemented an internationally ...
Innovate Research. Innovate Research stands as a leading Contract Research Organization (CRO) dedicated to providing comprehensive services in the dynamic field of clinical research, established in January 2014 With a strategic presence across the globe, our offices ar... 💻 Website ↗ 📞 +91 78277 58840 View all details.
Synchron is a leading contract research organization in India, designed to meet the specific requirements of the clients in the field of : Clinical registration of new pharmaceutical products and medical devices, from phase I to phase IV, including Bioequivalence, Bioavailability, Pharmacokinetic / Pharmacodynamic studies, Statistical Analysis and Data Management strictly adhering to ICH and ...
Bangalore Office. As one of the leading Indian mid-size clinical CROs. Biosite guides clients from clinical development planning to a regulatory filing, providing flexible solutions whilst ensuring budget and deadlines are met and reducing clients' risks and delays.
Welcome to Xplora Clinical Research Services Pvt. Ltd. (XCRS) We are keen to introduce ourselves as a fast growing Contract Research Organization (CRO) with inbuilt Site Management Organization (SMO), started in the year 2012. A team of clinical research professionals with more than 10 years experience in handling trial related activities in ...
Founded in 1997, Novotech is a global full-service clinical Contract Research Organization (CRO) focused on partnering with biotech companies to accelerate the development of advanced and novel therapeutics at every phase. Recognized for its industry-leading contributions, Novotech has received numerous prestigious awards, including the CRO Leadership Award 2023, and the Cell & Gene Therapy ...
SIRO - India's top CRO for clinical research services, operations, consulting, pharmacovigilance, and trials management. Trusted in Indian clinical research. ... END TO END CLINICAL RESEARCH ORGANIZATION. One stop shop for services ranging from medical writing and clinical operations, to data management and biostatistics.
To list your clinical trial on CTRI, we can assist you with CTRI Registration and CTRI submission over the CTRI online portal. Clinical Trials in India - Get Local / Global Clinical Research Services, CRO industry. For CRO in pharma services or Contract Research Organization in Delhi, Call +91 7672005050.
The top 15 CROs globally would be in the following order based on 2023 revenue: LabCorp (Covance), with 15.05 billion USD (2023) and 75000 employees (Labcorp, 2024). In February 2015, Labcorp completes its $6 billion purchase of Covance, Inc., creating the world's leading health care diagnostics company.
It commenced with a vision and dream of Mr. Onkar Kanwar, the Chairman of Apollo Tyres Ltd and Artemis Medicare Services Limited, to build a world class research center with cutting edge technology, state-of-the-art infrastructure and best of the scientific minds promoting basic and translational research to enhance the quality of patient care ...
Ophthalmology. Since 2010, Syneos Health has executed 290+ ophthalmology projects, including 35 Wet AMD studies, and developed and commercialized 48 approved eye care products, making us a leading ophthalmology contract research organization providing clinical development and commercialization services for drugs, devices and diagnostics in ...
Parexel is proudly among the world's largestclinical research organizations. A dedicated CRO providing the full range of Phase I to IV clinical development services and leveraging the breadth of our clinical, regulatory and therapeutic expertise, our team of more than 21,000 global professionals works in partnership with biopharmaceutical ...
Gurgaon: Gurugram University has taken a significant step towards providing students with advanced education in clinical research through its partnership with Artemis Hospital. The collaboration ...
Clinical Research & Data Management in Gurgaon - Get best price quotes from Clinical Research Organization, Clinical Data Management also download Clinical Research & Data Management contact addresses and phone numbers to your mobile from Sulekha.
3+ years of relevant clinical research and TMF experience at a biotechnology, pharmaceutical company or CRO required. ... Experience in a pharmaceutical or clinical research organization. Familiarity with regulatory requirements and guidelines for clinical trial documentation.
The eCourse in Clinical Research Methods is intended to support professional development through an eLearning mode. The course will provide greater flexibility to those with constraints in terms of time, distance, or other reasons. Further, self-paced learning using adult learning technique spread over a duration of four months will be less ...